This set of Class 11 Biology Chapter 9 Multiple Choice Questions & Answers (MCQs) focuses on “Chemical Analysis – 3” for Medical Entrance Exams.
1. Which of the following acids is used for the chemical analysis of living tissues?
a) Trichloroacetic acid
b) Trifluoroacetic acid
c) Tribromoacetic acid
d) Triiodoacetic acid
View Answer
Explanation: Trichloroacetic acid is used for the chemical analysis of living tissues like a vegetable or a piece of liver, etc. and the tissue is grinded using a mortar and a pestle.
2. What are biomolecules?
a) Molecules of quantum mechanics
b) Molecules of Life
c) Molecules related to string theory
d) It is a molecule
View Answer
Explanation: Biomolecules means the molecules of life or all the carbon compounds that we get from living tissues can be called as biomolecules.
3. What is the percentage weight of carbon in the earth’s crust?
a) 0.01
b) 0.3
c) 0.03
d) 0.14
View Answer
Explanation: The percentage weight of carbon in the earth’s crust is 0.03. It is 18.5 in the human body. The percentage weight of hydrogen in earth’s crust is 0.14 and in the human body is 0.5.
4. What is the use of elemental analysis?
a) To add those nutrients in food
b) To find the elemental composition of living tissues
c) To gain confidence
d) To research the unknown
View Answer
Explanation: Elemental analysis gives the elemental composition of living tissues in the form of hydrogen, oxygen, chlorine, carbon, etc. while analysis for compounds gives an idea of the kind of organic and inorganic constituents present in living tissues.
5. Why do we use dry weights for the chemical analysis?
a) Because it is vulnerable
b) Because of water leaks everywhere
c) Because water is not absorbed by chemicals
d) Because different organisms have different water-absorbing capacities
View Answer
Explanation: During a chemical analysis, one weighs a small amount of living tissue, say a leaf or liver and this weight is known as wet weight. After that, we dry it. All the water evaporates. The remaining material gives dry weight. Now because different organisms have different water-holding capacities, so their wet weights would be different.
6. Amino acids contain an amino group and an acidic group at different carbons
a) True
b) False
View Answer
Explanation: Amino acids are organic compounds containing an amino group and an acidic group as substituents on the same carbon i.e., the α carbon. Hence, they are called α-amino acids.
7. What is the other name of α-amino acids?
a) Substituted methanes
b) Substituted alcohols
c) Substituted alkyl halides
d) Substituted carboxylic acids
View Answer
Explanation: α-amino acids are also called as substituted methanes. Four substituent groups are occupying the four valency positions. These are hydrogen, carboxyl group, amino group and a variable R group.
8. Based on which of the following there are different kinds of amino acids?
a) Nature of R group
b) Nature of amino group
c) Nature of carboxyl group
d) Nature of hydrogen
View Answer
Explanation: Based on the nature of the R group there are many amino acids. However, those which occur in proteins are only of twenty types. The R group can be hydrogen, alcohol, etc.
9. What is the name of the given structure?
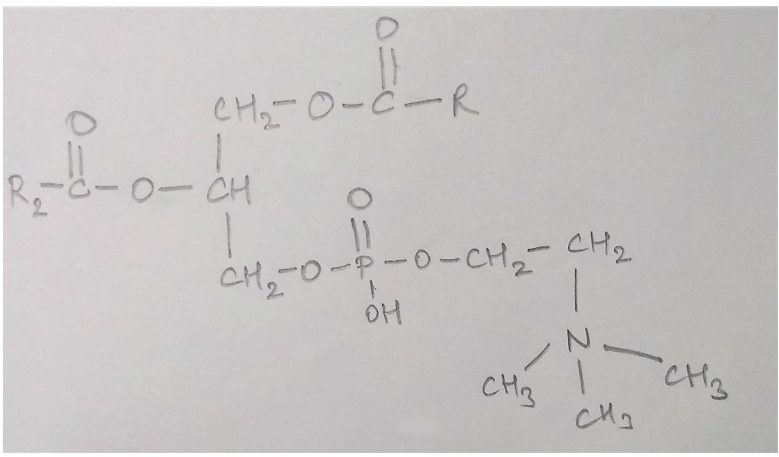
a) Glucose
b) Arabinose
c) Phospholipid
d) Uridine
View Answer
Explanation: The given structure depicts a phospholipid known as lecithin. These lipids have phosphorous and a phosphorylated organic compound in them. Lecithin is found in the cell membrane of every cell and on the surface of lung alveoli.
10. Which of the following is a basic amino acid?
a) Tryptophan
b) Tyrosine
c) Lysine
d) Glutamic acid
View Answer
Explanation: Lysine and arginine are some of the basic amino acids. Tryptophan, tyrosine and phenylalanine are aromatic amino acids. Aspartic acid and glutamic acid are some examples of acidic amino acids.
11. What is the name of the given structure?
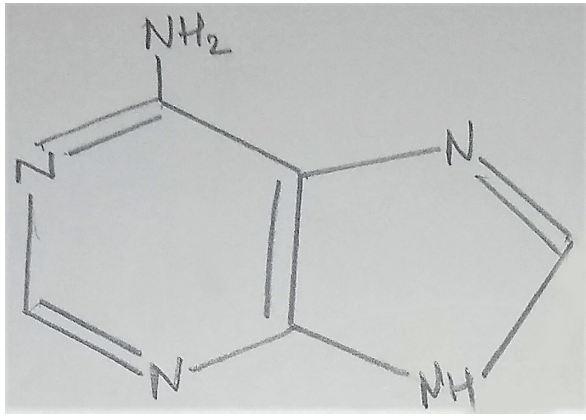
a) Adenosine
b) Adenine
c) Adenylic acid
d) Uridine
View Answer
Explanation: The given structure represents adenine. Living organisms have several carbon compounds in which heterocyclic rings can be found. Some of these are nitrogen bases- adenine, guanine, cytosine, uracil and thymine.
12. Lipids are generally water-soluble.
a) True
b) False
View Answer
Explanation: Lipids are generally water – insoluble. They could be simple fatty acids. A fatty acid has a carboxyl group attached to an R group. The R group could be a methyl, ethyl or any higher number of carbon – containing groups.
13. How many carbon atoms are there in palmitic acid?
a) 16
b) 18
c) 20
d) 22
View Answer
Explanation: Palmitic acid has 16 carbon atoms including carboxyl carbon. Arachidonic acid has 20 carbon atoms including the carboxyl carbon. These fatty acids could be saturated or unsaturated.
14. Which of the following is not a nucleotide?
a) Adenylic acid
b) Thymidylic acid
c) Guanylic acid
d) Cytidine
View Answer
Explanation: Adenylic acid, thymidylic acid, guanylic acid, uridylic acid and cytidylic acid are nucleotides. Adenosine, guanosine, thymidine, uridine and cytidine are nucleosides.
15. What is the name of the given structure?
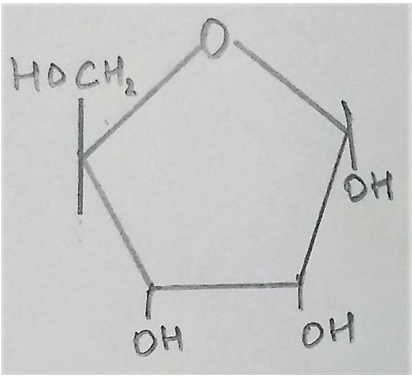
a) Ribose
b) Glucose
c) Deoxyribose
d) Fructose
View Answer
Explanation: The given structure represents ribose. Sugars like ribose and deoxyribose when are found attached to a nitrogen base, they are called nucleosides.
More Biology MCQ for Medical Entrance Exams:
- Biology MCQ for Medical Entrance Exam (Set 2)
- Biology MCQ for Medical Entrance Exam (Set 3)
- Biology MCQ for Medical Entrance Exam (Set 4)
To practice all chapters and topics of class 11 Biology, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Practice Class 11 - Mathematics MCQs
- Practice Class 11 - Chemistry MCQs
- Check Class 11 - Books
- Practice Class 12 - Biology MCQs
- Practice Class 11 - Physics MCQs
