This set of Class 12 Chemistry Chapter 12 Multiple Choice Questions & Answers (MCQs) focuses on “Aldehydes and Ketones Chemical Reactions – 2”.
1. Catalytic hydrogenation of _______ gives propan-2-ol.
a) formaldehyde
b) acetaldehyde
c) propionaldehyde
d) acetone
View Answer
Explanation: Reduction of acetone in the presence of catalysts like Ni, Pt or Pd converts it into propan-2-ol, which is a secondary alcohol. It can also be reduced by the reaction with LiAlH4 or NaBH4. Aldehydes on similar reaction give primary alcohols.
2. What is the product of Clemmensen reduction on acetophenone?
a) Benzaldehyde
b) Methyl benzene
c) Ethyl benzene
d) Benzophenone
View Answer
Explanation: The carbonyl group of acetophenone is reduced to CH2 group and results in the formation of a hydrocarbon when subjected to Clemmensen reduction, i.e., reaction with Zn-Hg and concentrated HCl.
3. A compound on treatment with hydrazine followed by heating up to 473K with KOH in ethylene glycol gives propane. Identify the compound.
a) Methanal
b) Ethanal
c) Propanal
d) Acetone
View Answer
Explanation: Aldehydes and ketones are reduced to respective hydrocarbons when treated with hydrazine followed by heating with KOH/NaOH in high boiling solvents. This is known as Wolff-Kishner reduction. The CO group of acetone is reduced to CH2, and propane is obtained.
4. Acetaldehyde is oxidised to ______ in the presence of nitric acid.
a) methanoic acid
b) ethanoic acid
c) acetone
d) ethanol
View Answer
Explanation: Aldehydes are easily oxidised to carboxylic acids with same number of carbon atoms, in the presence of oxidising agents like HNO3, KMnO4, etc. This is because of the presence of H atom next to CO group, which can be easily converted to OH group with any bond cleavage.
5. What are the major products of the oxidation of pentan-2-one with concentrated HNO3 at very high temperatures?
a) Butanoic acid and methanoic acid
b) Propanoic acid and ethanoic acid
c) Pentanoic acid
d) Butanoic acid and ethanoic acid
View Answer
Explanation: Ketones undergo oxidation accompanied by C-C bond cleavage to give a mixture of carboxylic acids having lesser number of carbon atoms than the parent ketone. In case of petan-2-one (which is unsymmetrical), the cleavage occurs such that the CO group stays with the smaller alkyl group (Popoff’s rule). Hence, the bond between C-2 and C-3 will be broken to give a mixture of ethanoic acid and propanoic acid.
6. Four compounds A, B, C and D were heated individually with Tollen’s reagent. It was found that all compounds other than D formed a silver mirror on the inside of their test tubes. Identify the compound D.
a) Acetaldehyde
b) Butyraldehyde
c) Acetone
d) Benzaldehyde
View Answer
Explanation: When aldehydes are heated with ammoniacal silver nitrate solution (Tollen’s reagent), it reduces the silver ions to metallic silver and forms a silver mirror. Ketones do not give this test as they are different to oxidise.
7. Which of the following is Tollen’s reagent?
a) Ammoniacal silver nitrate solution
b) Aqueous copper sulphate
c) Alkaline sodium potassium tartarate
d) Mixture of sodium carbonate, sodium citrate and Cu2+ complex
View Answer
Explanation: Tollen’s reagent is prepared by adding ammonium hydroxide solution to silver nitrate solution till the grey precipitate of Ag2O first formed just gets dissolved.
8. What is the proportion of Fehling solution A to Fehling solution B in the solution of Fehling’s reagent for conducting Fehling’s test?
a) 1:1
b) 1:2
c) 2:1
d) 1:3
View Answer
Explanation: Two solutions, aqueous copper sulphate and alkaline sodium potassium tartarate are combined in equal proportions to result in Fehling’s solution. This is an important reagent in the conduction of Fehling’s test.
9. When an aldehyde is heated with Fehling’s solution, a reddish-brown precipitate is formed due to which compound?
a) CuS
b) AgBr
c) Cu2O
d) CdS
View Answer
Explanation: Fehling’s reagent has Cu2+ ions which oxidise aldehyde by adding an O atom and turning it into corresponding carboxylate ion. In this process, a reddish-brown compound is precipitated due to the formation of cuprous oxide.
10. Which of the following compounds can be distinguished from iodoform test?
a) Benzaldehyde and benzophenone
b) Benzaldehyde and formaldehyde
c) Acetophenone and acetaldehyde
d) Acetophenone and benzophenone
View Answer
Explanation: Iodoform test involves the reaction of a compound with sodium hypoiodite to detect the presence of CH3CO or CH3CH(OH) group. A positive test forms CHI3 (iodoform) in the form of a yellow precipitate. Acetaldehyde and acetophenone give yellow ppt in this test, whereas formaldehyde, benzophenone and benzaldehyde do not undergo this test due to the absence of CH3CO group in their structures.
11. Which of the following does not give yellow ppt on iodoform test?
a) Propan-2-ol
b) Butan-2-ol
c) Pentan-2-one
d) Pentan-3-one
View Answer
Explanation: Pentan-2-one consists of a CH3CO groups and gives positive for iodoform test. Propan-2-ol and butan-2-ol pass iodoform test because of the presence of CH3CH(OH) group which gets oxidised to CH3O group during reaction. On the other hand, pentan-3-one consists of only ethyl groups on either side of the carbonyl carbon and is not oxidised by sodium hypoiodite to give iodoform.
12. Which of the following compound does not undergo aldol condensation?
a) Acetaldehyde
b) Propanal
c) Propanone
d) Benzaldehyde
View Answer
Explanation: Aldehydes and ketones that have at least one α-hydrogen atom undergo aldol condensation, where the α-hydrogen of one molecule becomes attached to the carbonyl carbon of the second molecule to form aldols and ketols respectively.
13. How many products are formed from the aldol condensation reaction between ethanal and propanal?
a) 1
b) 2
c) 3
d) 4
View Answer
Explanation: Ethanal and propanal both contain α-hydrogen atoms and undergo aldol condensation to form a mixture of four products. The self-aldol products of ethanal and propanal are but-2-enal and 2-methylpent-2-enal respectively. The two products formed from the cross-aldol condensation between one molecule of ethanal and one molecule of propanal are 2-methylbut-2-enal and pent-2-enal.
14. Identify the product A of the following reaction.
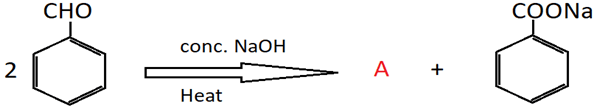
a) Phenol
b) Benzyl alcohol
c) Acetophenone
d) Benzophenone
View Answer
Explanation: Benzaldehyde does not contain any α-hydrogen and undergoes self- oxidation and reduction on treatment with conc. NaOH. One molecule of it is oxidised to carboxylic acid salt, and the other molecule is reduced to an alcohol (A). This is known as Cannizzaro’s reaction.
15. Formaldehyde cannot undergo aldol condensation as well as Cannizzaro’s reaction.
a) Trues
b) False
View Answer
Explanation: Formaldehyde (HCHO) does not contain an α-hydrogen and hence cannot undergo self-condensation to form aldol product. However, it undergoes Cannizzaro’s reaction to given methanol and potassium formate.
Sanfoundry Global Education & Learning Series – Chemistry – Class 12.
To practice all chapters and topics of class 12 Chemistry, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Practice Class 11 - Chemistry MCQs
- Practice Class 12 - Physics MCQs
- Practice Class 12 - Mathematics MCQs
- Practice Class 12 - Biology MCQs
- Check Class 12 - Chemistry Books
