This set of Class 12 Chemistry Chapter 13 Multiple Choice Questions & Answers (MCQs) focuses on “Preparation of Amines – 2”.
1. Which of the following reagents cannot be used to convert ethanenitrile to ethylamine?
a) H2/Ni
b) LiAlH4
c) Na(Hg), ethanol
d) Sn, HCl
View Answer
Explanation: Nitriles on reduction with lithium aluminium hydride or catalytic hydrogenation or with Na(Hg)-ethanol produce primary amines.
2. How many more carbon atoms are present in the amine formed from the reduction of a nitrile, than in the nitrile itself?
a) 0
b) 1
c) 2
d) 1 or 2
View Answer
Explanation: The -CN group of the nitrile is reduced to -CH2-NH2 group. This means that the parent carbon chain of the amine has one more carbon than the parent chain of the nitrile. However, the total number of carbons in both compounds is same as one of the carbon in nitriles is present in the cyanide group.
3. The reduction of phenyl isocyanide with H2 and Ni catalyst gives a/an _________
a) primary amine
b) secondary amine
c) tertiary amine
d) arylalkyl amine
View Answer
Explanation: The reduction of isocyanide compounds (where the CN group is attached through N atom) with H2/Ni gives secondary amines or N-alkyl amines. For example, phenyl isocyanide gives N-methylaniline.
4. What is the product of the following reaction?
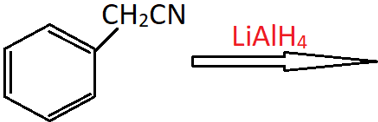
a) Phenylmethanamine
b) 1-Phenylethan-1-amine
c) 2-Phenylethan-1-amine
d) Toluene
View Answer
Explanation: Benzyl cyanide reacts with LiAlH4 to undergo reduction of the CN group to a CH2NH2 group and form a primary aromatic amine. The formula of this product will be C6H5CH2CH2NH2, which is 2-phenylethan-1-amine.
5. Which of the following amines cannot be formed from the reduction of amides with LiAlH4?
a) Ethylamine
b) Benzenamine
c) Benzylamine
d) Ethylmethylamine
View Answer
Explanation: The -CONH2 group of amides is reduced to a -CH2NH2 group and results in a compound having the same number of carbon atoms. The simplest aromatic amide is benzamide, which is reduced to give benzylamine. Hence, aniline cannot be formed as there is no corresponding amide that can be reduced to it with LiAlH4. However, it can be achieved by Hoffmann bromamide degradation.
6. Only primary amines can be obtained from the reduction of amides with LiAlH4.
a) True
b) False
View Answer
Explanation: Primary, secondary or tertiary amines can be formed depending on whether the amide used is primary, secondary or tertiary. This is because irrespective of the number of alkyl groups, the CO group of amides is directly reduced to CH2.
7. Ethyldimethylamine is obtained from the reduction of _______ with LiAlH4.
a) acetamide
b) benzamide
c) N-methylacetamide
d) N,N-dimethylacetamide
View Answer
Explanation: Ethyldimethylamine is a tertiary amine with two methyl substituents directly attached to N atom. It can be obtained only from the reduction of a corresponding tertiary amide that has two methyl substituents attached to N atom.
8. Which path gives propan-1-amine as the product in the reaction below?
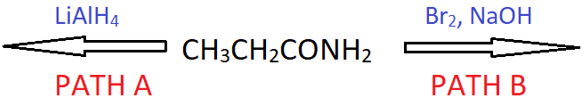
a) Only A
b) Only B
c) Both A and B
d) Not A nor B
View Answer
Explanation: Propanamine has three carbon atoms and so does the parent amide. So, the reduction of CO to CH2 will give the required compound. Whereas in path B, the amide undergoes Hoffmann bromamide degradation to give ethanamine.
9. Which of the following is not a by product of Hoffmann bromamide degradation of acetamide with alcoholic KOH?
a) KBr
b) KCN
c) K2CO3
d) H2O
View Answer
Explanation: Acetamide (CH3CONH2) reacts with Br2 and 4 molecules of KOH to form methylamine (main product) along with 2 molecules KBr, 2 molecules of H2O and one molecule of K2CO3.
10. Hoffmann bromamide degradation reaction is used for preparing _______ amines.
a) primary
b) secondary
c) tertiary
d) mixed
View Answer
Explanation: Mixed amines are a classification of secondary and tertiary amines. The Hoffmann bromamide degradation is only possible with 1° amides so as to produce primary amines.
11. The best reagent for converting 2-phenylpropanamide to 1-phenylethanamine is ________
a) H2/Ni
b) Na(Hg)/C2H5OH
c) LiAlH4
d) NaOH/Br2
View Answer
Explanation: 2-Phenylpropanamide has 9 carbon atoms while 1-phenylethamine has 8 carbon atoms. This depicts stepping down of carbon from amide to amine conversion and this takes place in Hoffmann degradation reaction.
12. Which of the following compounds undergoes Hoffmann bromamide degradation reaction?
a) C6H5NH2
b) C6H5NO2
c) C6H5CONH2
d) C6H5CH2NH2
View Answer
Explanation: Only primary amides undergo this reaction with bromine in NaOH to form primary amines. Benzamide undergoes this reaction to form benzenamine or aniline.
13. Which of the following amines can be prepared from Gabriel phthalimide synthesis?
a) Benzylamine
b) Aniline
c) o-Toluidine
d) N-Methylbenzenamine
View Answer
Explanation: Since Gabriel phthalimide synthesis is a nucleophilic substitution reaction, aryl halides cannot be prepared form this. This is because of the sp2 hybridised aryl carbon and stable benzene ring.
14. Gabriel phthalimide synthesis can be used for preparing phenylmethanamine.
a) True
b) False
View Answer
Explanation: Potassium phthalimide can undergo nucleophilic substitution with a benzyl halide to form N-phenylphthalimide, which on hydrolysis with NaOH gives benzylamine or phenylmethanamine.
15. Which of the following steps is not present in Gabriel phthalimide synthesis?
a) Treating phthalimide with alcoholic KOH
b) Heating potassium phthalimide with alkyl halide
c) Alkaline hydrolysis of N-alkylphthalimide
d) Heating phthalic acid with NaOH
View Answer
Explanation: The chemical compounds involved in the Gabriel phthalimide synthesis are phthalimide, potassium phthalimide and N-alkyl phthalimide. These are formed in order when reacted with different reagents like KOH and haloalkanes.
Sanfoundry Global Education & Learning Series – Chemistry – Class 12.
To practice all chapters and topics of class 12 Chemistry, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Check Class 12 - Chemistry Books
- Practice Class 12 - Physics MCQs
- Practice Class 11 - Chemistry MCQs
- Practice Class 12 - Mathematics MCQs
- Practice Class 12 - Biology MCQs
