This set of Class 12 Chemistry Chapter 12 Multiple Choice Questions & Answers (MCQs) focuses on “Methods of Carboxylic Acids Preparation”.
1. Which of the following is known as Jones reagent?
a) KMnO4 in alkaline medium
b) CrO3 in H2SO4
c) K2Cr2O7 in acidic medium
d) KMnO4 in H2SO4
View Answer
Explanation: Chromium trioxide in an aqueous solution with sulphuric acid in known as Jones reagent. It Is an important compound in the preparation of carboxylic acids from alcohols.
2. Benzoic acid is obtained from the oxidation of _______ with alkaline KMnO4 followed by treatment with mineral acid.
a) phenol
b) benzaldehyde
c) acetophenone
d) benzyl alcohol
View Answer
Explanation: Only primary alcohols undergo oxidation in the presence of common oxidising agents followed by reaction with H3O+ to give the respective carboxylic acids. This reaction is proceeded by the removal of both hydrogen atoms from the alpha carbon and formation of a double bond between C and O.
3. Identify the most suitable reagent for the conversion of ethanal to acetic acid.
a) Alkaline KMnO4; H3O+
b) Jones reagent
c) Tollen’s reagent
d) LiAlH4
View Answer
Explanation: Aldehydes are easily oxidized to carboxylic acids having the same number of carbon atoms as the parent aldehyde, when reacted even with mild oxidising agents like Tollen’s reagent.
4. Identify the product B in the reaction chain shown.
![]()
a) Ethanoic acid
b) Propanoic acid
c) Butanoic acid
d) 2-Methylpropanoic acid
View Answer
Explanation: Ethyl bromide on reaction with alc. KCN gives ethyl cyanide by nucleophilic substitution. Nitriles can be hydrolysed to give amides and on further heating give a carboxylic acid which contains one more carbon than the original ethyl bromide. Hence, the acid contains three C atoms, making it propanoic acid.
5. Identify X in the following conversion.
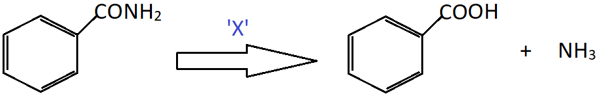
a) Alkaline KMnO4; H3O+
b) KOH; heat
c) H3O+; heat
d) KMnO4-KOH; heat
View Answer
Explanation: Amides undergo hydrolysis in the presence of heat to give corresponding carboxylic acids along with ammonia gas. In the reaction shown, benzamide undergoes oxidation to form benzoic acid.
6. Benzoic acid can be prepared by oxidation of tert-Butyl benzene.
a) True
b) False
View Answer
Explanation: tert-Butyl benzene consists of a tertiary group with no benzylic H. It is highly stable and does not undergo oxidation even under drastic conditions, to give aromatic carboxylic acids.
7. Which of the following pairs do not give the same compound on heating with alkaline potassium permanganate?
a) Toluene and propyl benzene
b) Toluene and n-Butyl benzene
c) Propyl benzene and isopropyl benzene
d) o-Xylene and n-Butyl benzene
View Answer
Explanation: All mono-substituted alkyl benzenes, with primary or secondary alkyl groups, on vigorous oxidation give benzoic acid. The entire side chain is oxidised irrespective of the length. In case of o-xylene, there are two methyl groups on the benzene ring and both of them are oxidised to carboxyl group, resulting in phthalic acid.
8. p-Xylene on reaction with acidified potassium dichromate at high temperature gives ________
a) benzoic acid
b) phthalic acid
c) terephthalic acid
d) no reaction
View Answer
Explanation: p-Xylene is a dimethyl substituted benzene which on vigorous oxidation, gets oxidised to an aromatic dicarboxylic acid, with the two carboxyl groups at para positions with respect to each other. This compound is called terephthalic acid.
9. Which of the following cannot be converted to benzoic acid on reaction with KMnO4-KOH followed by H3O+?
a) Ethyl benzene
b) Acetophenone
c) 4-Methylacetophenone
d) Styrene
View Answer
Explanation: The ethyl group, acetyl group and ethene group are all oxidised to potassium carboxylate groups which are further oxidised to carboxyl groups. In the case of 4-Methylacetophenone, there are two groups that will be oxidised to carboxyl groups, hence forming terephthalic acid instead of benzoic acid.
10. How can methyl magnesium bromide be converted to propanoic acid?
a) Jones reagent
b) KMnO4-KOH; heat
c) H3O+; heat
d) CO2-dry ether; H3o+
View Answer
Explanation: Methyl magnesium bromide (Grignard reagent) on reaction with CO2 forms an addition product containing an additional C carbon atom. This is decomposed in the presence of mineral acid to form propanoic acid.
11. 3-Chlorophenyl magnesium bromide on reaction with dry ice followed by acidification in mineral acid gives _________
a) 3-Chlorophenol
b) 3-Chlorophenylethanoic acid
c) 3-Chlorobenzaldehyde
d) 3-Chlorobenzoic acid
View Answer
Explanation: The MgBr group of 3-Chlorophenyl magnesium bromide (Grignard reagent) will be substituted by COOH group in the above reaction, to give a halogen substituted aromatic carboxylic acid.
12. Benzoic ethanoic anhydride on hydrolysis gives __________
a) benzoic acid and methanoic acid
b) benzoic acid and ethanoic acid
c) phenylethanoic acid and methanoic acid
d) no products
View Answer
Explanation: Benzoic ethanoic anhydride (C6H5COOCOCH3) is easily hydrolysed with water to give its corresponding acids, benzoic acid (by adding H to C6H5COO) and ethanoic acid (by adding OH to COCH3).
13. Ethanoyl chloride on hydrolysis with aqueous NaOH gives _______
a) acetate ion
b) acetic acid
c) propanoic acid
d) no reaction
View Answer
Explanation: Acid chlorides are readily hydrolysed with aqueous base to give carboxylate ions which further give corresponding carboxylic acids on acidification. It can be directly obtained by direct hydrolysis with water.
14. The final product(s) of basic hydrolysis followed by acidification of ethyl butanoate is _______
a) ethanoic acid
b) butanoic acid
c) ethanoic acid and butanoic acid
d) butanoic acid and ethanol
View Answer
Explanation: Ethyl butanoate (CH3CH2CH2COOC2H5) on basic hydrolysis forms CH3CH2CH2COONa and ethanol. Then this sodium carboxylate compound gets acidified to give butanoic acid.
15. Acidic hydrolysis of ethyl benzoate directly gives benzoic acid.
a) True
b) False
View Answer
Explanation: Acidic hydrolysis of esters directly gives carboxylic acids. In this case, the ethyl group of the ester is separated out as ethanol along with the main product, benzoic acid.
Sanfoundry Global Education & Learning Series – Chemistry – Class 12.
To practice all chapters and topics of class 12 Chemistry, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Practice Class 12 - Mathematics MCQs
- Practice Class 12 - Physics MCQs
- Practice Class 12 - Biology MCQs
- Practice Class 11 - Chemistry MCQs
- Check Class 12 - Chemistry Books
