This set of Class 12 Chemistry Chapter 12 Multiple Choice Questions & Answers (MCQs) focuses on “Nomenclature and Structure of Carbonyl Groups – 1”.
1. Which of the following compounds do not contain a carbonyl group?
a) Alcohol
b) Aldehyde
c) Ketone
d) Carboxylic acid
View Answer
Explanation: Carbonyl groups are those which contain a carbon-oxygen double bond. In alcohols, the bond between C and O is a single bond and hence does not contain a carbonyl group.
2. How many carbon atoms does formaldehyde have?
a) 0
b) 1
c) 2
d) 3
View Answer
Explanation: Formaldehyde is the common name of the simplest aldehyde, methanal. Its formula is HCHO and only one carbon which is also a part of the carbonyl group.
3. What is the common name of butanal?
a) n-Butanaldehyde
b) α-Butanaldehyde
c) n-Butyraldehyde
d) α-Butyraldehyde
View Answer
Explanation: Butanal is a straight chain aldehyde with 4 carbon atoms including the carbonyl group. There is no substituent present in the carbon chain and hence the prefix n- is given in the common name.
4. What is the common name of the compound which has a CHO group attached to the sp2 hybridised carbon of a benzene ring?
a) Benzanal
b) Benzaldehyde
c) Benzenecarbaldehyde
d) Phthaldehyde
View Answer
Explanation: The IUPAC name of the simplest aromatic aldehyde carrying the CHO group on a benzene ring is benzenecarbaldehyde. Its common name is benzaldehyde, which is also accepted by IUPAC.
5. Which of the following is the incorrect name for the following compound?
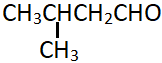
a) 3-Methylbutanal
b) β-Methylbutyraldehyde
c) γ-Methylbutyraldehyde
d) Isovaleraldehyde
View Answer
Explanation: The compound is a 4-carbon chain aldehyde with a methyl group substituted at the beta-carbon position, i.e., the carbon next to the carbon directly linked to the aldehyde group.
6. The prefix valer- is used for generally used for naming aldehydes with how many carbon atoms in the structure?
a) 3
b) 4
c) 5
d) 6
View Answer
Explanation: When aldehydes have exactly 5 carbon atoms in their structure, including the carbonyl group and any substituted groups, the aldehyde is given the prefix valer- in the common naming system. For example, pentanal is named as n-Valeraldehyde.
7. What is the correct IUPAC naming of the compound shown?
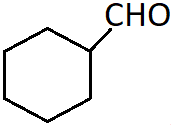
a) Benzenecarbaldehyde
b) Cyclohexanal
c) Cyclohexyl aldehyde
d) Cyclohexanecarbaldehyde
View Answer
Explanation: When the CHO group is attached to a ring, the suffix carbaldehyde is added after the name of the cycloalkane. The numbering of the ring carbons starts form the carbon attached to the aldehyde group.
8. Identify the correct IUPAC name of CH3-CH=CH-CHO.
a) But-2-enal
b) 2-Butenal
c) Buten-2-al
d) Butenal
View Answer
Explanation: The carbon atom of the CHO group is assigned the first number. So, in this case, the double bond occurs at second carbon and hence, the prefix 2- is used for the naming of double bond -en.
9. The compound 3-Phenylprop-2-enal is also known as _________
a) Crotonaldehyde
b) Cinnamaldehyde
c) Salicylaldehyde
d) Vanillin
View Answer
Explanation: In cinnamaldehyde, a benzene ring is attached to the end carbon of the aldehyde chain and also has a carbon double bond at the alpha carbon. Its formula is C6H5-CH=CH-CHO.
10. Identify the correct common name for the following compound.
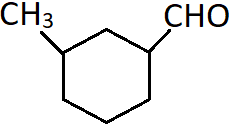
a) 3-Methylcyclohexanecarbaldehyde
b) 3-Methylbenzecarbaldehyde
c) 3-Methylbenzaldehyde
d) γ-Methylcyclohexanecarbaldehyde
View Answer
Explanation: This compound is a cyclic aldehyde where numbering starts from the carbon to which the CHO group is attached. So, the carbon to which methyl group is attached is the third carbon. In the common system, this carbon is known as the gamma carbon.
11. What is the IUPAC name of the sown compound?
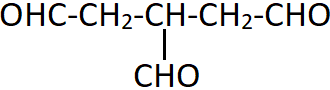
a) Propane-1,2,3-trial
b) 3-Formylpentan-1,5-dial
c) 2-(Formylmethyl)butan-1,4-dial
d) Propane-1,2,3-tricarbaldehyde
View Answer
Explanation: There are three aldehydic groups present in the given compound and none of them have preference over the other. The CHO groups are considered as substituents with the parent chain containing only three carbon atoms. This is done to give identical treatment to all the aldehydic groups.
12. How many aldehydic groups does the compound phthaldehyde have?
a) 1
b) 2
c) 3
d) 4
View Answer
Explanation: Phthaldehyde is an aromatic compound containing two CHO groups, one on each adjacent carbon of the benzene ring. To give equal importance to both the aldehydic groups, it is named as benzene-1,2-dicarbaldehyde in the IUPAC system.
13. The compound 3-Bromobenzaldehyde is named as γ-Bromobenzaldehyde in the common system.
a) True
b) False
View Answer
Explanation: The prefix gamma- is used for substituents in a straight carbon chain. For aromatic substituted compounds, the prefixes ortho-, meta- and para- are used. Hence, 3-Bromobenzaldehyde is named as m-Bromobenzaldehyde in the common system.
14. Vanillin is an aldehydic compound.
a) True
b) False
View Answer
Explanation: Vanillin consists of a benzene ring substituted with CHO group and OH group on opposite side. Additionally, there is a methoxy group at meta position with respect to the aldehydic group. Its IUPAC name is 4-Hydroxy-3-methoxybenzaldehyde.
Sanfoundry Global Education & Learning Series – Chemistry – Class 12.
To practice all chapters and topics of class 12 Chemistry, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Practice Class 11 - Chemistry MCQs
- Practice Class 12 - Mathematics MCQs
- Check Class 12 - Chemistry Books
- Practice Class 12 - Biology MCQs
- Practice Class 12 - Physics MCQs
