This set of Class 12 Chemistry Chapter 13 Multiple Choice Questions & Answers (MCQs) focuses on “Diazonium Salts Chemical Reactions – 3”.
1. The reaction involving the conversion of diazonium salts to azo compounds is known as _______ reaction.
a) diazotisation
b) deamination
c) coupling
d) carbylamine
View Answer
Explanation: Benzenediazonium salts react with highly reactive electron rich aromatic compounds to form azo compounds of the form Ar-N=N-Ar, where Ar represents an aromatic group. This type of reaction is known as coupling reaction.
2. The coupling reaction is a type of _________ reaction.
a) electrophilic addition
b) electrophilic substitution
c) nucleophilic addition
d) nucleophilic substitution
View Answer
Explanation: During a coupling reaction, the diazonium cation which has a positive charge on the terminal nitrogen acts as the electrophile, and the electron rich compounds act as nucleophiles.
3. The coupling reaction of benzenediazonium chloride with aniline occurs in a ______ medium.
a) strongly basic
b) weakly basic
c) neutral
d) weakly acidic
View Answer
Explanation: Aniline is a compound that is slightly basic in nature. To counter the basic effect of aniline during its coupling with diazonium salt, in is conducted in a slightly acidic medium of pH 4 to 5.
4. The reaction between benzenediazonium chloride and phenol results in a ______ coloured compound.
a) yellow
b) orange
c) red
d) purple
View Answer
Explanation: Benzenediazonium chloride undergoes coupling with phenol to form an azo compound, p-hydroxyazobenzene. This compound is orange in colour.
5. The reaction between a diazonium salt and aniline in a slightly acidic medium gives _______
a) o-aminoazobenzene
b) m-aminoazobenzene
c) p-aminoazobenzene
d) no reaction
View Answer
Explanation: Diazonium salts undergo coupling with aniline to form p-aminoazobenzene. The coupling predominantly occurs at the para position to the amino group.
6. What is the most suitable pH of the medium for the conduction of coupling reaction of benzenediazonium chloride with phenol?
a) 2
b) 4
c) 7
d) 10
View Answer
Explanation: The coupling reaction with phenol occurs in a basic medium of pH value approximately 9 to 10. This is to counter the acidic nature of phenol and as a result produce water (by the combination of OH– and H+) during the reaction.
7. Identify the colour of the compound ‘A’ from the following reaction.
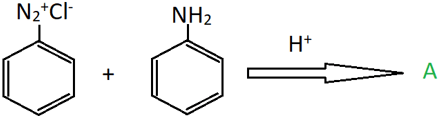
a) Yellow
b) Colourless
c) Green
d) Blue
View Answer
Explanation: Diazonium salts on reaction with aniline form azo compounds which are coloured. This is because of the complex system involving the N-N double bond between two aromatic rings. In this case, compound A is p-aminobenzene, which is yellow coloured.
8. The reaction between benzenediazonium chloride and N,N-dimethylaniline, in an acidic medium at 273 K gives an orange coloured compound.
a) True
b) False
View Answer
Explanation: This reaction yields p-dimethylaminoazobenzene, which is a yellow solid commonly known as methyl yellow. However, in an aqueous solution at low pH, methyl yellow appears red in colour.
9. When a diazonium salt is treated with p-cresol, at which position with respect to the hydroxy group does the coupling occur?
a) ortho
b) meta
c) para
d) coupling does not take place
View Answer
Explanation: Coupling generally occurs para to the hydroxy group. However in this case, the para position is preoccupied by CH3 group (cresol), and as a result the coupling takes place at ortho position.
10. What is the product of the following reaction?
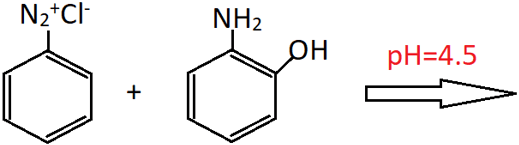
a) 2-Amino-4-phenylazophenol
b) 6-Amino-2-phenylazophenol
c) 2-Amino-5-phenylazophenol
d) 2-Amino-3-phenylazophenol
View Answer
Explanation: The reactant (o-aminophenol) consists of both hydroxy and amino groups. Since the reaction takes place in an acidic medium, the coupling occurs at para position (first preferred) with respect to the amino group. If the reaction was to take place in a basic medium, the coupling would have occurred at para position with respect to hydroxy group.
11. p-Aminophenol on coupling with benzenediazonium chloride in a basic medium gives 4-amino-3-phenylazophenol.
a) True
b) False
View Answer
Explanation: As the reaction takes place in a basic medium, the coupling will take place at the ortho position (since para position is already occupied by amino group) with respect to OH group. This results in the compound 4-amino-2-phenylazophenol.
Sanfoundry Global Education & Learning Series – Chemistry – Class 12.
To practice all chapters and topics of class 12 Chemistry, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Practice Class 11 - Chemistry MCQs
- Practice Class 12 - Biology MCQs
- Practice Class 12 - Physics MCQs
- Practice Class 12 - Mathematics MCQs
- Check Class 12 - Chemistry Books
