This set of Class 12 Chemistry Chapter 13 Multiple Choice Questions & Answers (MCQs) focuses on “Amines Nomenclature – 1”.
1. Which of the following is the IUPAC name of the compound in which one hydrogen of ammonia is replaced by an ethyl group?
a) Ethylamine
b) Aminoethane
c) Ethanamine
d) Ethane amine
View Answer
Explanation: Ethylamine and aminoethane are the names of CH3CH2NH2 according to the common system and second system respectively. In the IUPAC system, the naming is done by replacing the ‘e’ of the alkane by amine.
2. Which of the following names of amines belong strictly to the common system?
a) Ethylmethylamine
b) Aniline
c) Benzenamine
d) Propan-1-amine
View Answer
Explanation: In the common system, aliphatic amines are named by prefixing the alkyl group to amine, i.e., alkylamine. In case of two different alkyl groups, it is named by listing the alkyl groups in alphabetical order before the word amine, just like in ethylmethylamine. Aniline is a common name but is also accepted by IUPAC.
3. Which of the following names of aromatic amines does not belong to the common naming system?
a) Aniline
b) o-Toluidine
c) 4-Bromoaniline
d) N–Methylaniline
View Answer
Explanation: In the common system, the aromatic amines have special names derived from the basic compound aniline and depending upon the type of substituent group. The prefixes o-, m- and p- are used to depict the position of the substituent in common system. Whereas, the number position of carbon is used in the IUPAC system.
4. What is the correct common name of H2N-CH2-CH2-NH2?
a) Ethylenediamine
b) 1,2-Diaminoethane
c) Ethyldiamine
d) Aminoethylamine
View Answer
Explanation: In this compound, more than one amino group is present at different positions in the parent chain. Both the amino groups are given equal importance and none of them is considered a substituent on the other. The prefix di- is used before amine, and since it is a disubstituted ethane, it is referred to as ethylene.
5. What is the correct IUPAC name of H2N-(CH2)5-NH2?
a) Pentan-1,5-diamine
b) 1,5-Diaminopentane
c) Pentamethylenediamine
d) Pentane-1,5-diamine
View Answer
Explanation: The parent carbon chain consists of five atoms with one amino group at each end, i.e., first and fifth carbon. The prefix di is used to indicate the two amino groups and the letter ‘e’ of the suffix part of pentane is retained. Hence, the name pentane-1,5-diamine.
6. In the IUPAC system, the prefix N– is generally used for _______ amines.
a) primary
b) secondary
c) tertiary
d) secondary and tertiary
View Answer
Explanation: For the naming of 2° and 3° amines, the locant N is used as a prefix to identify the substituent attached to the nitrogen atom. In case of tertiary amines with same substituent groups, the prefix used is N, N–.
7. Identify the incorrect name of the shown compound.
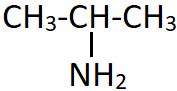
a) N-Propylamine
b) Isopropylamine
c) 2-Aminopropane
d) Propan-2-amine
View Answer
Explanation: The shown compound is a primary amine and hence the locant N cannot be used as it is invalid. The amino group is attached to the second carbon of the propyl group (which is also known as isopropyl substituent).
8. Which of the following is the correct IUPAC name of (CH3)3N?
a) Trimethylamine
b) N-Methylethanamine
c) N,N-Dimethylmethanamine
d) N,N,N-Trimethylamine
View Answer
Explanation: The compound is a tertiary amine with three methyl substituents. One of the methyl groups is considered as the parent chain with the N atom and the other two acts as substituents. These are represented by N, N- as they are the same groups and are directly attached to the nitrogen atom.
9. Benzylamine is the IUPAC name of aniline.
a) True
b) False
View Answer
Explanation: Aniline is an aromatic aryl amine, whereas benzylamine is am arylalkyl amine. Aniline is both accepted as the common and IUPAC names. The IUPAC naming of aryl amines is done by replacing the suffix ‘e’ of the arene by amine. Therefore, aniline is named as benzenamine.
10. What is the correct IUPAC name of CH3-NH-CH2CH3?
a) N-Methylethanamine
b) N-Ethylmethanamine
c) N-Ethyl-N-methylmethanamine
d) N-Ethyl-N-methylamine
View Answer
Explanation: This is a secondary amine with an ethyl and a methyl group. The smaller alkyl group (CH3) is considered as the substituent which is attached to the N atom. Hence, it is N-methyl and not N-ethyl.
11. What is the IUPAC name of the following compound?
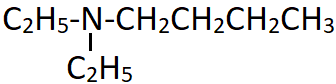
a) N-Ethylpentan-1-amine
b) N-Ethylpentan-2-amine
c) N,N-Diethylbutan-1-amine
d) N-Ethyl-N-Butylethanamine
View Answer
Explanation: This is a tertiary amine with two identical alkyl substituents (ethyl group) and one butyl group which acts as the major chain. The prefix N,N-Di is used for the ethyl groups and this collective group is attached at the first carbon of the butyl group.
12. The IUPAC name of allylamine is prop-2-en-1-amine.
a) True
b) False
View Answer
Explanation: Allylamine (NH2-CH2-CH=CH2) is an unsaturated amine with the amino group attached to an allyl carbon. In such compounds, the amino group is given preference and hence the double bond lies at the second carbon.
13. Identify the incorrect name of the aromatic primary amine with the formula C7H9N.
a) Benzenamine
b) Benzylamine
c) Phenylaminomethane
d) Phenylmethanamine
View Answer
Explanation: Since it is an aromatic compound, the phenyl group C6H5 has to present with CH4N. Since it is a primary amine, the group NH2 can be separated leaving us with CH2. So, the formula of the compound is C6H5CH2NH2, which is benzylamine. Benzenamine has the formula C6H5NH2.
14. Hexamethylenediamine is named as __________ in the IUPAC system.
a) Hexane-1,3-diamine
b) Hexane-1,4-diamine
c) Hexane-1,5-diamine
d) Hexane-1,6-diamine
View Answer
Explanation: The formula is NH2-(CH2)6-NH2 which is the chain of 6 carbon atoms and two amino groups at the ends of the chain, i.e., first and sixth carbon. This type of substitution is named and hexamethylene.
Sanfoundry Global Education & Learning Series – Chemistry – Class 12.
To practice all chapters and topics of class 12 Chemistry, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Practice Class 12 - Mathematics MCQs
- Practice Class 12 - Physics MCQs
- Check Class 12 - Chemistry Books
- Practice Class 11 - Chemistry MCQs
- Practice Class 12 - Biology MCQs
