This set of Class 12 Chemistry Chapter 13 Multiple Choice Questions & Answers (MCQs) focuses on “Amines Chemical Reactions – 1”.
1. Amines are generally ______ in nature.
a) electrophilic
b) acidic
c) basic
d) neutral
View Answer
Explanation: Amines behave as nucleophiles due to the presence of unshared pair of electrons on N atom. This also makes them proton acceptor, and react with acids to form salts.
2. Which of the following is associated with decrease in pKb value of amines?
a) Increase in acidic strength
b) Increase in basic strength
c) Better proton donation
d) Better electron acceptor
View Answer
Explanation: The basic strength of amines is expressed in terms of dissociation constant Kb. Greater the Kb value, stronger the base. It is more commonly expressed as pKb=-logKb. Hence smaller the value of pKb, more is the basic strength of the amine.
3. The equilibrium constant of the reaction of amines with _____ is taken as a measure of its basic character.
a) an acid
b) a base
c) water
d) a Lewis base
View Answer
Explanation: For the reaction,
RNH2 + H2O ↔ RNH3+ + OH–, the dissociation constant is,
Keq = ([RNH3+][OH–])/([RNH2][H2O])
\(\Rightarrow\) Keq[H2O] = ([RNH3+][OH–])/[RNH2]
\(\Rightarrow\) Kb = ([RNH3+][OH–])/[RNH2]
4. What is the product formed when ethanamine reacts with HBr?
a) NH4Br
b) CH3NH3Br
c) CH3CH2NHBr
d) CH3CH2NH3Br
View Answer
Explanation: Since amines are basic in nature, they react with acids to form salts of ammonium. Ethanamine (CH3CH2NH2) reacts with HBr in a reversible reaction to form ethylammonium bromide through addition.
5. The reaction between methylamine and hydrogen iodide results in the formation of a _______
a) colourless liquid
b) dark coloured gas
c) white solid
d) yellow liquid
View Answer
Explanation: Methylamine and hydrogen iodide are allowed to mix at very cold temperatures for about 2 hours. The resulting product is allowed to evaporate and methanaminium iodide (CH3NH3I) is obtained in the form of a white powder.
6. Which of the following is not produced on the reaction of methylammonium chloride with sodium hydroxide?
a) HCl
b) CH3NH2
c) H2O
d) NaCl
View Answer
Explanation: Amine salts on treatment with a base like NaOH gives back the parent amine along with a water molecule and a salt. For example, CH3NH3Cl with NaOH gives methanamine , water and sodium chloride.
7. Identify X and Y respectively in the following reaction.
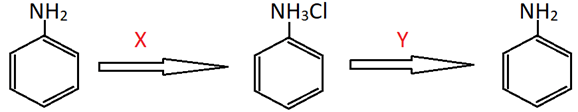
a) OH–, HCl
b) HCl, OH–
c) H2O, OH–
d) HCl, H2O
View Answer
Explanation: Aniline undergoes addition in the presence of HCl to give anilinium chloride (salt) which on action with OH– ions (from a base like NaOH) gives back aniline.
8. What is the correct order of pKb values of the following amines?
a) Methanamine > Ethanamine > Benzenamine
b) Benzenamine > Ethanamine > Methanamine
c) Ethanamine > Methanamine > Benzenamine
d) Benzenamine > Methanamine > Ethanamine
View Answer
Explanation: The higher pKb value means lower basic strength. This means that benzenamine has the lower basic strength among methanamine and ethanamine. This is because of the electron withdrawing nature of the aryl group. Also in primary amines, the increase in size of alkyl group increases +I effect leading to high electron density on N atom and as a result higher basicity.
9. The treatment of N,N-dimethylaniline with acetic acid gives no reaction.
a) True
b) False
View Answer
Explanation: N,N-Dimethylaniline reacts with acetic acid to form a salt, N,N-dimethylanilinium acetate. This is due to the basic nature of amines.
10. If the pKb value of ammonia is 4.75, predict the pKb value of methanamine?
a) 3.38
b) 4.70
c) 8.92
d) 9.38
View Answer
Explanation: Aliphatic amines are stronger bases than ammonia and hence have a much lower pKb than ammonia. This is because of the electron donating nature of alkyl groups which increase the negative charge on nitrogen atom, making it less prone to electron acceptance.
11. If the Kb values of ammonia, methylamine and ethylamine are x, y and z respectively, identify the correct relation between x, y and z from the following.
a) x > y
b) y < z
c) x > z
d) x > y > z
View Answer
Explanation: Larger the value of Kb, stronger is the base. Since ethylamine is a stronger base than methylamine which is stronger than ammonia, due to the effect of alkyl groups, the Kb vale of methylamine will be less than that of ethylamine (y<z).
12. Consider three gaseous alkylamines A, B and C or 1°, 2° and 3° respectively. What will be the correct order of their basicity?
a) A > B > C
b) C > B > A
c) B > A > C
d) B > C > A
View Answer
Explanation: In the gaseous state, the solvation effect is missing and hence the expected order of basicity will be 3° > 2° > 1°. This is because as the number of alkyl groups increase, the +I effect strengthens and more negative charge is accumulated on the N atom. This makes the unpaired share mire available for sharing with the proton of the acid.
13. The basic strength of alkylamines does not depend on which of the following?
a) Number or alkyl groups
b) Size of alkyl groups
c) Physical state of the amine
d) Presence of an aromatic ring
View Answer
Explanation: Since only basicity of alkylamines is in question the presence of aromatic ring is not considered. As the size and number of alkyl groups increases, the stability of ammonium ion (formed from the amine) increases due to dispersal of more positive charge by the +I effect of alkyl groups. The physical state, gaseous or aqueous, also determines the basicity as the hydration effect comes into play.
14. What is the correct order of basicity of aliphatic amines purely on the basis of solvation effect of the ammonium cation?
a) 1° > 2° > 3°
b) 3° > 2° > 1°
c) 2° > 1° > 3°
d) 2° > 3° > 1°
View Answer
Explanation: In aqueous phase, the greater the size of the ion, lesser will be the hydration and less stabilised is the ion. Since the stability of ions is directly proportional to the basic strength of amines, primary amines will be the most basic and tertiary amines will be the least basic. This order is completely opposite to that based on the inductive effect of alkyl groups.
15. Stearic hinderance of alkyl groups has an effect on the basic character of amines.
a) True
b) False
View Answer
Explanation: When the alkyl group is small, there is no stearic hinderance to hydrogen bonding. But when the alkyl group size or number increases, there will be hinderance to formation of hydrogen bonds, and this affects the order of basic strength of amines.
Sanfoundry Global Education & Learning Series – Chemistry – Class 12.
To practice all chapters and topics of class 12 Chemistry, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Check Class 12 - Chemistry Books
- Practice Class 12 - Biology MCQs
- Practice Class 11 - Chemistry MCQs
- Practice Class 12 - Mathematics MCQs
- Practice Class 12 - Physics MCQs
