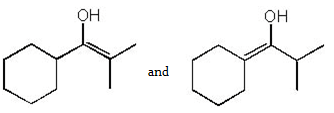This set of Organic Chemistry Multiple Choice Questions & Answers (MCQs) focuses on “Tautomerism”.
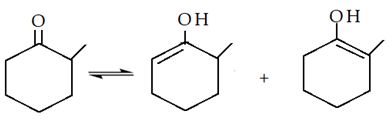
1. How many enolisable hydrogens are there in the following compound?
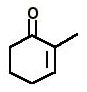
a) 4
b) 5
c) 6
d) 7
View Answer
Explanation: The hydrogen atom present on the carbon which is adjacent to the carbon attached to the functional group, and here it is 4.
2. How many tautomers can you draw for the following ketone?
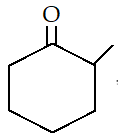
a) 1
b) 2
c) 3
d) 4
View Answer
3. How many tautomers can you draw for the following ketone?
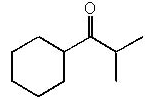
a) 1
b) 2
c) 3
d) 4
View Answer
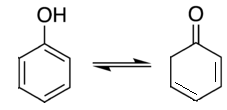
4. Which of the following is a tautomer of phenol?
a) 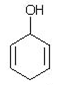
b) 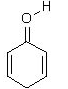
c) 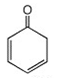
d) 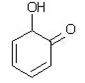
View Answer
5. Which of the following compound exhibit tautomerism?
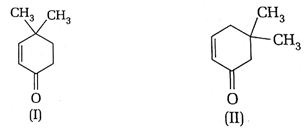
a) I
b) II
c) I and II
d) Neither I nor II
View Answer
6. In keto-enol from presence of which type of hydrogen is must?
a) Alpha
b) Beta
c) Gamma
d) Any position of hydrogen
View Answer
Explanation: For Keto-Enol tautomerism, keto form should have α-hydrogen, as shown in below example.
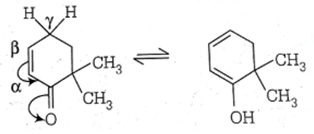
7. Which of the following tautomeric form is more stable?
CH3-CH=O ↔ CH2=C-OH
(I) (II)
a) I
b) II
c) Both are equally stable
d) Both are unstable
View Answer
Explanation: The keto–enol tautomerization chemical equilibrium is highly thermodynamically driven, and at room temperature the equilibrium heavily favors the formation of the keto form.
8. What is the stability order for the following compounds?
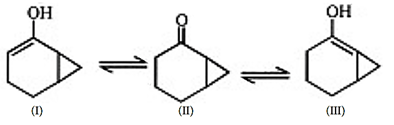
a) I > II > III
b) II > I > III
c) III > II > I
d) I > III > II
View Answer
Explanation: The keto–enol tautomerization, less stable enolate ion will be formed faster as it is thermodynamically stable, so III is more stable than I.
II is most sable as it is keto form.
9. What is the stability order for the following compounds?
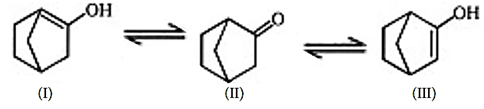
a) I > II > III
b) II > I > III
c) III > II > I
d) I > III > II
View Answer
Explanation: The keto–enol tautomerization, less stable enolate ion will be formed faster as it is thermodynamically stable, so III is more stable than I.
II is most sable as it is keto form.
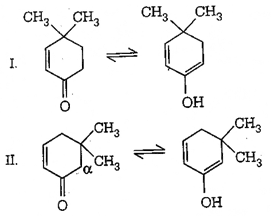
10. Which of the following structure can show tautomerism?
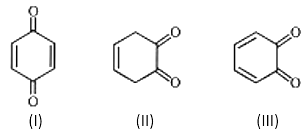
a) I
b) II
c) III
d) I and III
View Answer
Explanation: Quinone has an α-hydrogen; however, it is a vinylic hydrogen (hydrogen attached to a carbon atom which is sp2 hybridized), it is very difficult to abstract such a hydrogen, and hence it becomes very less acidic. Therefore, no abstraction of α-hydrogen from it, by a base, hence, it does not show tautomerism, same is for III.
Sanfoundry Global Education & Learning Series – Organic Chemistry.
To practice all areas of Organic Chemistry, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Practice Chemical Engineering MCQs
- Apply for Chemical Engineering Internship
- Check Chemical Engineering Books
- Check Organic Chemistry Books