This set of Organic Chemistry Interview Questions and Answers for Experienced people focuses on “Reaction Mechanism of Organic Compounds”.
1. Why are aryl halides less reactive towards nucleophilic substitution reactions as compared to alkyl halides?
a) The formation of a less stable carbanion
b) Longer carbon halogen bond
c) The inductive effect
d) Sp2-hybridized carbon attached to the halogen
View Answer
Explanation: Overlapping of sp2 orbital of carbon with p-orbital of halogen is one of the reasons. Due to conjugation double bond character in alkyl halide.
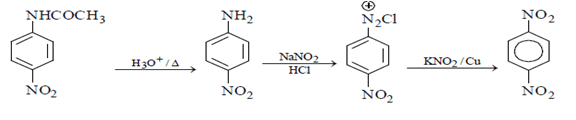
2. What will be the (X) in the below mentioned reaction sequence?
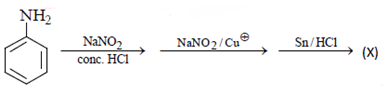
a)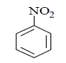
b) 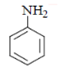
c) 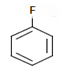
d)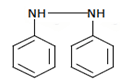
View Answer
Explanation: As we can see in below reaction, that firstly diazotisation reaction will occur. This is followed by reaction of NaNO2 1 with diazonium ion forming nitro benzene, which will undergo reduction and form aniline.
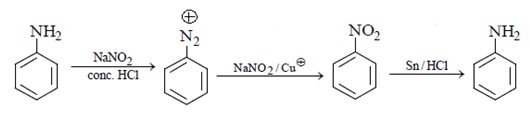
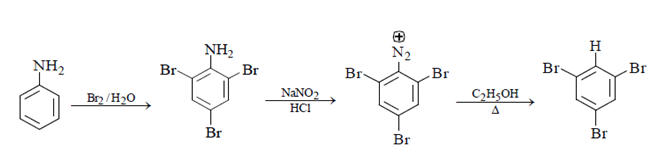
3. In the following reaction sequence, what will be X?
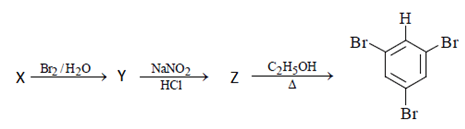 .
.
a) Benzoic acid
b) Salicylic acid
c) Phenol
d) Aniline
View Answer
Explanation: Aniline will be X as we can see below reaction. Free bromination will occur, and bromine will get add to ortho and para position followed diazotisation and a rapid reaction will take place between diazonium ion and C2H5OH and 1, 3, 5-tribromo benzene will form.
4. What will be the final product in the below reaction?
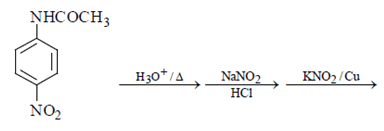
a) 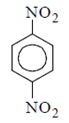
b) 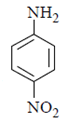
c) 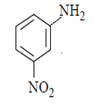
d) 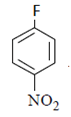 .
.
View Answer
Explanation: As we can see hydrolysis is the first step which will form a amine group containing compound. This amino-compound undergoes diazotisation and a rapid reaction will take place between diazonium ion and KNO2 and the ‘a’ product will form.
5. Which of the following structures represent the correct major product for the below reaction?

a)
b)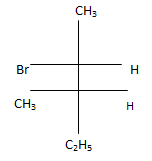
c)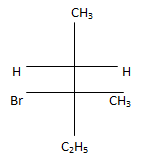
d)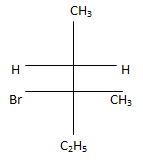 .
.
View Answer
Explanation: According to following mechanism we can say that some are major products. As we can see protonation of hydroxyl group followed by dehydration will leads to hydride shift to the adjacent positively charged carbon for formation of more stable carbocation. This will unleash two possibility of attack by Br- at the new carbocation from upward and backward and two products will be formed that are shown below.
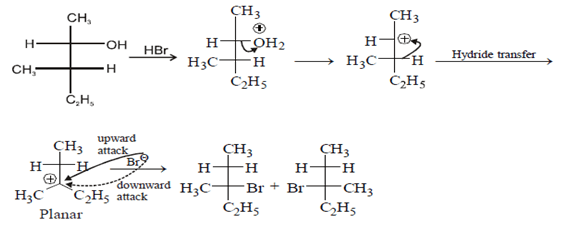
6. Which of the following is not true for SN1 reactions?
a) They occur through a single step concerted reaction
b) They are favoured by polar solvents
c) Tertiary alkyl halides generally react through this mechanism
d) Concentration of nucleophile does not affect the rate of such reactions
View Answer
Explanation: SN1 reaction is a two-step reaction, step one is the leaving group leaves, and the second step is the substrate forms a carbocation intermediate. Formation of carbocation intermediate is the rate determining step. Since for the formation of stable intermediate carbocation, highly polar solvent is required. The bulky substituents prevent the nucleophiles from approaching the carbon, which is attached directly to the halogen. SN1is also more favorable as the neighboring alkyl groups are electron donating, which helps to stabilize the carbocation. SN1 reaction because the nucleophile is not a part of the rate-determining step.
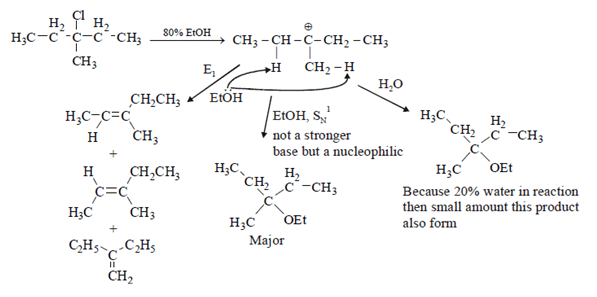
7. What is not true about below reaction?
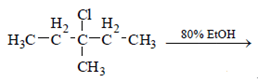 .
.
a) Major product is given by SN1 reaction
b) Through E1 mechanism 3 alkenes are formed
c) 3-Methylpentane-3-ol is also formed as one of the product
d) Fractional distillation of elimination product will give two fractions
View Answer
Explanation: As we see below mechanism major product is formed by SN1 mechanism and E1 mechanism forms 3 alkenes. 3-Methylpentane-3-ol is also formed in the reaction. Fractional distillation of elimination product will give three fractions. Hence statement d is false.
8. What will be the total number of isomers formed when 2-methyl butane is subjected to monochlorination?
a) 5
b) 4
c) 3
d) 6
View Answer
Explanation: Monochlorination of 2 methyl butane gives two pairs of enantiomers. 1st pair of enantiomer is 1-chloro-2-methyl butane and 2nd pair of enantiomer is 2-chloro-3-methyl butane.
1-chloro-3-methyl butane and 2-chloro-2-methyl butane products are also formed.
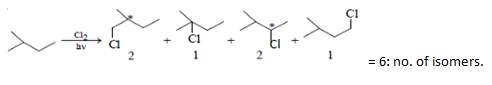
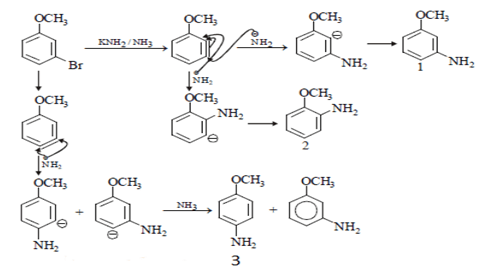
9. The number of substitution products formed when metabromo anisole is treated with KNH2/NH3?
a) 1
b) 2
c) 3
d) 4
View Answer
Explanation: Nucleophile (-NH2)will attack on Br and there will be three possibilities of bond formation and those are ortho, para and meta position as shown in the below structures number 2, 3 and 1 respectively.
10. How many number of pie e- is present in benzene?
a) 6
b) 7
c) 8
d) 9
View Answer
Explanation: Pie electron are those which show de-localization of the electrons, in same plan or p orbitals and that there aren’t alternating double and single bonds. But the electron in one p-p overlapping is present perpendicular to the plane of ring.
Sanfoundry Global Education & Learning Series – Organic Chemistry.
To practice all areas of Organic Chemistry for Interviews, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Check Organic Chemistry Books
- Check Chemical Engineering Books
- Practice Chemical Engineering MCQs
- Apply for Chemical Engineering Internship
