This set of Organic Chemistry Multiple Choice Questions & Answers (MCQs) focuses on “Stereoisomers”.
1. What is the complete IUPAC name of the following substance?
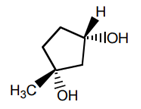
a) (1S,3S)-1-methylcyclopentane-1,3-diol
b) (1R,3R)-1-methylcyclopentane-1,3-diol
c) (1S,3R)-1-methylcyclopentane-1,3-diol
d) (1R,3S)-1-methylcyclopentane-1,3-diol
View Answer
Explanation: The IUPAC name will be (1R,3S)-1-methylcyclopentane-1,3-diol, where 1 position shows R and 3 position shows S.
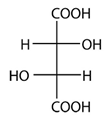
2. How many stereoisomers are there for the following structure?
a) 1
b) 2
c) 3
d) 4
View Answer
Explanation: In the case of 2,3-dihydroxybutanedioic acid, known as tartaric acid, the two chiral centers have the same four substituents and are equivalent. As a result, two of the four possible stereoisomers of this compound are identical due to a plane of symmetry, so there are only three stereoisomeric tartaric acids.
3. How many stereoisomers of 3-bromo-2-butanol, CH3CH(OH)CHBrCH3, exist?
a) 3
b) 1
c) 2
d) 4
View Answer
Explanation: 3-Bromo-2-butanol has two chirality centers with different sets of four substituents. Each chirality center can have either an S or R configuration. For a compound with n chirality centers there is the possibility of a maximum of 2n stereoisomers. In this case there is more than one stereoisomer.
4. How many stereoisomers of 2,3-butanediol, CH3CH(OH)CH(OH)CH3, exist?
a) 3
b) 4
c) 1
d) 2
View Answer
Explanation: 2,3-Butanediol has two chirality centers but the sets of four substituents on each chirality center are identical. Each chirality center can have either an S or R configuration. The (2R,3R) and (2S,3S) compounds are enantiomers of one another, but the structures drawn as (2R,3S) and (2S,3R) compounds are identical (this is the achiral meso compound), so there are only three different stereoisomers. For a compound with n chirality centers there is the possibility of a maximum of 2n stereoisomers. However, if the sets of four substituents on each chirality center are identical there will be fewer than 2n stereoisomers.
5. What is the complete IUPAC name of the following stereoisomer?

a) (1S)-2-ethyl-3,3-dimethylcyclopentane
b) (1R)-2-ethyl-3,3-dimethylcyclopentane
c) (1S)-1-ethyl-,3,3-diimethylcyclopentane
d) (1S)-3-ethyl-1,1-diimethylcyclopentane
View Answer
Explanation: The IUPAC name will be (1S)-1-ethyl-,3,3-diimethylcyclopentane, where at 1 position it is S.
6. Which of the following structures represent the same stereoisomer?

a) Only 1 and 2
b) Only 2 and 3
c) 1, 2 and 3
d) Only 3 and 1
View Answer
Explanation: It is important that you can rotate around carbon-carbon single bonds to make different conformations, and to rotate molecules in three-dimensions to compare their chirality chemistry (model kits are often useful). Another way to determine which of these structures are identical is to determine the configuration of each chirality center. In this case, for each chirality center the priority of the substituents is -Cl >; -CHClCH3 >; -CH3 >; -H. For example, the configurations of the chirality centers of compound 1 are both S. However, 1 and 3 are not identical.
7. Which of the following compounds can exhibit geometrical isomerism?
a) 1-Hexene
b) 2-Methyl-2-Pentene
c) 3-methyl-1-pentene
d) 2-Hexene
View Answer
Explanation: There are several isomers of hexene, depending on the position and geometry of the double bond in the chain. One of the most common industrially useful isomers is 1-hexene an alpha-olefin.
8. How many number of stereoisomers possible for 2, 3-pentanediol?
a) 3
b) 4
c) 5
d) 6
View Answer
Explanation: 2, 3-pentanediol contains two chiral atoms. So, the number of possible stereoisomers is 4 (22).
9. How many chiral stereoisomers can be drawn for CH3CHFCHFCH(CH3)2?
a) 4
b) 5
c) 6
d) 7
View Answer
Explanation: CH3CHFCHFCH(CH3)2 contains 2 chiral atoms. So, the number of possible stereoisomers is 4 (22).
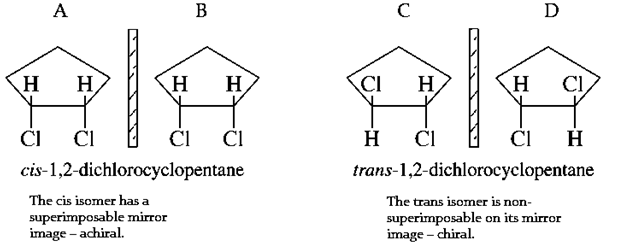
10. What is the total number of isomers, stereoisomers included, designated by the dichlorocyclopentane?
a) 7
b) 6
c) 4
d) 5
View Answer
Explanation: 3 structural isomers, but if you are considering chirality/diastereomers, there are two chiral carbons, so the maximum number of stereoisomers is 22=4, from which cis-1,2-dichlorocyclopentane and cis-1,3-dichlorocyclopentane will have same structure in their individual 4 isomers as shown below, so total isomers will be 7.
Sanfoundry Global Education & Learning Series – Organic Chemistry.
To practice all areas of Organic Chemistry, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Practice Chemical Engineering MCQs
- Check Chemical Engineering Books
- Check Organic Chemistry Books
- Apply for Chemical Engineering Internship
