This set of Thermodynamics Multiple Choice Questions & Answers (MCQs) focuses on “Entropy Generation in a Closed and Open System-1”.
1. The entropy of any closed system can increase in which if the following way?
a) by heat interaction in which there is entropy transfer
b) dissipative effects or internal irreversibilities
c) both of the mentioned
d) none of the mentioned
View Answer
Explanation: These two processes increase the entropy of a closed system.
2. Entropy increase dS of the system can be expressed as
a) dS=dS(due to external heat interaction)-dS(due to internal irreversibility)
b) dS=dS(due to external heat interaction)+dS(due to internal irreversibility)
c) dS=-dS(due to external heat interaction)-dS(due to internal irreversibility)
d) dS=-dS(due to external heat interaction)+dS(due to internal irreversibility)
View Answer
Explanation: Total entropy increase of the system is the sum of these two entropies.
3. The entropy increase due to internal irreversibility is also called entropy production or entropy generation.
a) true
b) false
View Answer
Explanation: This entropy is generated during the process within the system.
4. Which of the following statement is true?
a) if the isentropic process is reversible, it must be adiabatic
b) if the isentropic process is adiabatic, it cannot but be reversible
c) if the process is adiabatic and reversible, it must be isentropic
d) all of the mentioned
View Answer
Explanation: An adiabatic process need not be isentropic, since entropy can also increase due to friction.
5. Lost work is given by
a) pdV-dW
b) pdV+dW
c) -pdV-dW
d) pdV*dW
View Answer
Explanation: The lost work d(LW) indicates the work that is lost due to irreversibility.
6. The amount of entropy generation is given by
a) S2+S1+∫(dQ/T)
b) S2-S1+∫(dQ/T)
c) S2-S1-∫(dQ/T)
d) none of the mentioned
View Answer
Explanation: Here (S2-S1) is the entropy change of the system and ∫(dQ/T) is the entropy transfer.
7. Any thermodynamic process is accompanied by entropy generation.
a) true
b) false
View Answer
Explanation: This comes from the second law.
8. Which of the following statement is false?
a) for a reversible process, entropy generation is zero
b) the entropy generation does not depend on the path the system follows
c) for an irreversible process, entropy generation is greater than zero
d) none of the mentioned
View Answer
Explanation: Entropy generation is not a thermodynamic property and depends on the path that system follows.
9. If the path A causes more entropy generation than path B, then
a) path A is more irreversible than path B
b) path A involves more lost work
c) both of the mentioned
d) none of the mentioned
View Answer
Explanation: The amount of entropy generation quantifies the intrinsic irreversibility of the process.
10. In an open system, there is a transfer of which of the following quantity?
a) mass
b) energy
c) entropy
d) all of the mentioned
View Answer
Explanation: In an open system, there is a transfer of all these three quantities.
11. The rate of entropy increase of the control volume ____ or ____ the net rate of entropy transfer to it.
a) exceeds or is less than
b) exceeds, is equal to
c) is less than, or equal to
d) none of the mentioned
View Answer
Explanation: The difference is the entropy generated within the control volume due to irreversibility.
12. Mass and energy are conserved quantities, but entropy is generally not conserved.
a) true
b) false
View Answer
Explanation: This is a basic fact about entropy.
13. The rate at which entropy is transferred out must ____ the rate at which entropy enters the control volume.
a) be less than
b) equal to
c) exceed
d) none of the mentioned
View Answer
Explanation: The difference is the rate of entropy generated within the control volume owing to irreversibilities.
14. A chip dissipates 2 kJ of electric work and rejects it as heat transfer from its surface which is at 50°C to 25°C air. How much entropy is generated in the chip?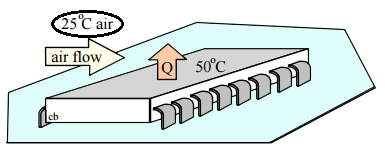
a) 4.19 J/K
b) 5.19 J/K
c) 6.19 J/K
d) 7.19 J/K
View Answer
Explanation: C.V.1 Chip with surface at 50°C, we assume chip state is constant.
U2 – U1 = 0 = 1Q2 – 1W2 = W(electrical,in) – Q(out,1)
S2 – S1 = 0 = – [Q(out,1)]/[T(surf)] + 1S2(gen1)
1S2(gen1) = [Q(out,1)]/[T(surf)] = W(electrical,in) / T(surf)
= 2/323.15 = 6.19 J/K.
15. A chip dissipates 2 kJ of electric work and rejects it as heat transfer from its surface which is at 50°C to 25°C air. How much entropy is generated outside the chip?
a) 0.419 J/K
b) 0.519 J/K
c) 0.619 J/K
d) 0.719 J/K
View Answer
Explanation: C.V.2 From chip surface at 50°C to air at 25°C, assume constant state.
U2 – U1 = 0 = 1Q2 – 1W2 = Q(out,1) – Q(out,2)
S2 – S1 = 0 = [Q(out,1) / T(surf)] – [Q(out,2) / T(air)] + 1S2(gen2)
1S2(gen2) = [Q(out,2) / T(air)] – [Q(out,1) / T(surf)] = (2/298.15) – (2/323.15) = 0.519 J/K.
Sanfoundry Global Education & Learning Series – Thermodynamics.
To practice all areas of Thermodynamics, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Check Chemical Engineering Books
- Practice Mechanical Engineering MCQs
- Check Mechanical Engineering Books
- Apply for Mechanical Engineering Internship
- Practice Chemical Engineering MCQs
