This set of Thermodynamics Questions & Answers for campus interviews focuses on “Second Law Efficiency-2”.
1. As Ta approaches Tr, second law efficiency
a) half
b) first law efficiency
c) zero
d) unity
View Answer
Explanation: The lower the Ta, lower will be second law efficiency, here Tr=source temperature and Ta=use temperature.
2. Which of the following statement explains the concept of energy cascading?
a) the fuel should first be used for high temperature applications
b) the heat rejected from these applications can then be cascaded to applications at lower temperatures
c) it ensures more efficient energy utilization
d) all of the mentioned
View Answer
Explanation: These are the main features of energy cascading.
3. Second law efficiency of different components can be expressed in different forms.
a) true
b) false
View Answer
Explanation: It is derived using the exergy balance rate.
4. A steam turbine inlet is at 1200 kPa and 500°C. The actual exit is at 300 kPa having an actual work of 407 kJ/kg. Find its second law efficiency?
a) 0.88
b) 0.98
c) 0.78
d) 0.68
View Answer
Explanation: To = 25°C = 298.15 K, hi = 3476.28 kJ/kg; si = 7.6758 kJ/kg K
he = hi – w(ac) = 3476.28 – 407 = 3069.28 kJ/kg
Te = 300°C; se = 7.7022 kJ/kg K
wrev = (hi – Tosi) – (he – Tose) = (hi – he) + To(se – si)
= (3476.28 – 3069.28) + 298.15(7.7022 – 7.6758)
= 407 + 7.87 = 414.9 kJ/kg
second law efficiency = w(ac)/w(rev) = 407 / 414.9 = 0.98.
5. A heat exchanger increases the availability of 3 kg/s water by 1650 kJ/kg by using 10 kg/s air which comes in at 1400 K and leaves with 600 kJ/kg less availability. What is the second law efficiency?
a) 0.625
b) 0.825
c) 0.925
d) 0.725
View Answer
Explanation: I = Φ(destruction) = Φ(in) – Φ(out) = 10 × 600 – 3 × 1650 = 1050 kW
second law efficiency = Φ(out)/Φ(in) = (3 × 1650)/(10 × 600)
= 0.825.
6. A heat engine receives 1 kW heat at 1000 K and gives out 600 W as work with the rest as heat to the ambient. Find the second law efficiency.
a) 0.655
b) 0.755
c) 0.855
d) 0.955
View Answer
Explanation: First law efficiency = 0.6/1 = 0.6
Φ(H) = [1-(To/Th)]Q = [1-(298.15/100)](1) = 0.702 kW
second law efficiency = 0.6/ 0.702 = 0.855.
7. A heat pump has a COP of 2 using a power input of 2 kW. Its low temperature is To and high temperature is 80°C, with an ambient at To. Find the second law efficiency.
a) 0.11
b) 0.41
c) 0.51
d) 0.31
View Answer
Explanation: Φ(H) = [1-(To/Th)]Q, Q = β*W = 2*2 = 4 kW
second law efficiency = Φ(H)/W = [1-(298.15/353.15)](4/2)
= 0.31.
8. The condenser in a refrigerator receives R-134a at 50°C, 700 kPa and it exits as saturated liquid at 25°C. The flow-rate is 0.1 kg/s and air flows in the condenser at ambient 15°C and leaving at 35°C. Find the heat exchanger second-law efficiency.
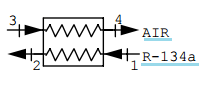
a) 0.77
b) 0.87
c) 0.47
d) 0.67
View Answer
Explanation: m1h1 + mah3 = m1h2 + mah4 (here m is the mass flow-rate)
ma = m1 × (h1 – h2)/(h4 – h3) = 0.1 × (436.89 – 234.59)/(1.004(35 – 15))
= 1.007 kg/s
ψ1 – ψ2 = h1 – h2 – T0(s1 – s2) = 436.89 – 234.59 – 288.15(1.7919 – 1.1201)
= 8.7208 kJ/kg
ψ4 – ψ3 = h4 – h3 – T0(s4 – s3) = 1.004(35 – 15) – 288.15 × 1.004 × ln (308.15/288.15)
= +0.666 kJ/kg
η(II) = ma(ψ4 – ψ3)/m1(ψ1 – ψ2) = 1.007(0.666)/[0.1(8.7208)] = 0.77.
9. Steam enters a turbine at 550°C, 25 MPa and exits at 5 MPa, 325°C at a flow rate of 70 kg/s. Determine the second law efficiency.
a) 0.68
b) 0.78
c) 0.88
d) 0.98
View Answer
Explanation: hi = 3335.6 kJ/kg, si = 6.1765 kJ/kg K,
he = 2996.5 kJ/kg, se = 6.3289 kJ/kg K
Actual turbine: w,ac = hi – he = 339.1 kJ/kg
Rev. turbine: w,rev = w,ac + T0(se – si) = 339.1 + 45.44 = 384.54 kJ/kg
η(II) = w,ac/w,rev = 339.1/384.54 = 0.88.
10. A compressor is used to bring saturated water vapour initially at 1 MPa up to 17.5 MPa, where the actual exit temperature is 650°C. Find the second-law efficiency.
a) 0.651
b) 0.751
c) 0.851
d) 0.951
View Answer
Explanation: hi = 2778.1 kJ/kg, si = 6.5864 kJ/kg K
Actual compressor: h(e,ac) = 3693.9 kJ/kg, s(e,ac) = 6.7356 kJ/kg K
-w(c,ac) = h(e,ac) – hi = 915.8 kJ/kg
i = T0[s(e,ac) – si] = 298.15 (6.7356 – 6.5864) = 44.48 kJ/kg
w(rev) = i + w(c,ac) = -915.8 + 44.48 = -871.32 kJ/kg
η(II) = -w(rev)/w(c,ac) = 871.32/915.8 = 0.951.
Sanfoundry Global Education & Learning Series – Thermodynamics.
To practice all areas of Thermodynamics for campus interviews, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Practice Chemical Engineering MCQs
- Check Mechanical Engineering Books
- Check Chemical Engineering Books
- Apply for Mechanical Engineering Internship
- Practice Mechanical Engineering MCQs
