This set of Mechanical Metallurgy Multiple Choice Questions & Answers (MCQs) focuses on “Creep & Stress Rupture – High Temperature Material Problem”.
1. The equilibrium number of vacancy in a crystal lattice ____________ with increasing temperature.
a) increases
b) decreases
c) remains constant
d) no relationship
View Answer
Explanation: The vibration of atoms increase with increase in the temperature; this causes the diffusion and movement of atoms easier. The energy of crystal increases, and to accommodate this energy, the number of vacancy increases.
2. At an elevated temperature, the material behaves as ____________
a) elastic
b) plastic
c) viscoelastic
d) viscous
View Answer
Explanation: At elevated temperature, the factor of time scale also comes into the picture. So the material under tensile loading or other kinds of load loses strength if kept for excessive time even below the limiting stress.
3. The creep becomes significant above _______ of the homologous temperature.
a) 0.2
b) 0.1
c) 0.5
d) 0.9
View Answer
Explanation: The creep becomes dominant above the temperature, which is half of the melting point of that material. If the homologous temperature is greater than 0.5, then creep becomes dominant.
4. The material at room temperature does not have any anelastic behavior.
a) True
b) False
View Answer
Explanation: Anelastic behavior is a delayed response in recovering the original dimension of the material. Even at the room temperature, the anelastic behavior is present, but the amount is minimal.
5. As the temperature increases, the anelastic behaviour of the material ________________
a) increases
b) decreases
c) remains constant
d) no relationship
View Answer
Explanation: As the temperature increases, the anelastic behavior of the material also increases. The creep is governed by the anelastic behavior of the material.
6. The elastic after effect is the decay of strain to zero with time after releasing the load on material.
a) True
b) False
View Answer
Explanation: The anelastic behavior of the material is termed as the delay in the strain recovery with time. This behavior is known as the elastic after effect.
7. If the material is expanded elastically with very high rate, such that there is no thermal equilibrium between the material and surrounding, and also the expansion is adiabatic (no exchange of heat). The temperature of the material with strain will __________
a) increase
b) decrease
c) remain constant
d) no relation
View Answer
Explanation: The variation of temperature of material with strain is given as:
-> dT/dε = -VmαET/cv
Where dT/dε = change in temperature with strain
Vm = Molar volume of the material
α = Coefficient of thermal expansion
E = Isothermal young’s modulus
T = Absolute temperature
Cv = specific heat at constant volume
So from the expression, it can be inferred that the change in temperature with strain is negative, Hence, the temperature will decrease.
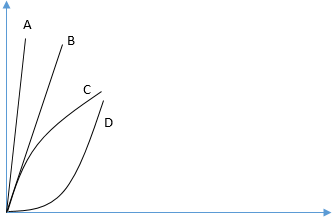
8. Two identical specimens are strained simultaneously in the elastic region. Sample-1 was loaded slowly and allowed to achieve thermal equilibrium, while sample-2 was loaded rapidly and heat transfer was not allowed. The stress-strain curve for these for sample 1 will be represented by the curve __________
a) A
b) B
c) C
d) D
View Answer
Explanation: The sample is loaded in the elastic region so that the curve will be linear, hence curve C and D not possible. Now when sample loaded is in thermal equilibrium, there will be no temperature rise in the sample so the modulus of the material will be lower than the one which is loaded rapidly. So answer will be curve-B.
9. Two identical specimens are strained simultaneously in the elastic region. Sample-1 was loaded slowly and allowed to achieve thermal equilibrium, while sample-2 was loaded rapidly and heat transfer was not allowed. The stress-strain curve for these for sample 2 will be represented by the curve __________

a) A
b) B
c) C
d) D
View Answer
Explanation: The sample is loaded in the elastic region so that the curve will be linear, hence curve C and D not possible. Now when sample loaded is without thermal equilibrium, there will be a temperature drop in the sample so the strength and modulus of the material will increase. Hence the curve A will represent the correct curve.
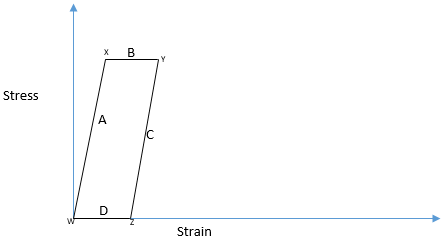
10. The following hysteresis cycle represents the adiabatic loading and unloading of a sample in the elastic region, without thermal equilibrium. Which part of the curve is associated with increase in the temperature of the sample?
a) A
b) B
c) C
d) D
View Answer
Explanation: First, the sample is loaded adiabatically and reached to point x. So the sample is cooled down below room temperature. If the time is allowed, the sample heats up and material is expanded and reaches to point Y. So the curve B is associated with increase in the sample temperature.
11. The following hysteresis cycle represents the adiabatic loading and unloading of a sample in the elastic region, without thermal equilibrium. Which part of the curve is associated with decrease in the temperature of the sample?

a) A
b) B
c) C
d) D
View Answer
Explanation: First, the sample is loaded adiabatically and reached to point x. So the sample is cooled down below room temperature. If the time is allowed, the sample heats up and material is expanded and reaches to point Y. So the curve B is associated with increase in the sample temperature. After this, when the sample is unloaded adiabatically, the temperature of the sample increases and reached to point Z. Hence, with time it cools down to room temperature and material shrinks. So, the curve D is associated with decreases in the temperature.
12. The following hysteresis cycle represents the adiabatic loading and unloading of a sample in the elastic region, without thermal equilibrium. Which region of the curve is associated with constant temperature?

a) A & C
b) B & D
c) C & D
d) D & A
View Answer
Explanation: The loading and unloading of the sample are sudden, so there is no temperature associated; hence, both the linear slope A & C represents the constant temperature.
Sanfoundry Global Education & Learning Series – Mechanical Metallurgy.
To practice all areas of Mechanical Metallurgy, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Practice Metallurgical Engineering MCQs
- Check Metallurgical Engineering Books
- Apply for Metallurgical Engineering Internship
- Check Mechanical Metallurgy Books
