This set of Engineering Materials & Metallurgy Multiple Choice Questions & Answers (MCQs) focuses on “Electrical Properties”.
1. What is the attribute of a material which resists the flow of electricity known?
a) Conductivity
b) Thermoelectricity
c) Dielectric strength
d) Resistivity
View Answer
Explanation: Resistivity is that attribute of a material which resists the flow of electricity, while conductivity is it’s reciprocal (or) opposite. The process by which two dissimilar metals join and produce a voltage is known as thermoelectricity. The dielectric strength of a material is its insulating capacity against voltage.
2. How is conductivity of a material defined?
a) ![]()
b) ![]()
c) ![]()
d) ![]()
View Answer
Explanation: Electrical conductivity is defined by
3. What is the electrical conductivity of Aluminum?
a) 6.3 * 107
b) 5.9 * 107
c) 3.5 * 107
d) 1 * 107
View Answer
Explanation: The electrical conductivity of Aluminum is 3.5 * 107. The electrical conductivity of Silver, Copper, and Iron is 6.3 * 107, 5.9 * 107, and 1 * 107 in that order. The electrical conductivity is generally measured at 20oC.
4. What is the electrical resistivity of Copper?
a) 1.59 * 10-8
b) 1.68 * 10-8
c) 2.65 * 10-8
d) 5.9 * 10-8
View Answer
Explanation: The electrical resistivity of Copper is 1.68 * 10-8. The electrical resistivity of Silver, Aluminum, and Zinc is 1.59 * 10-8, 2.65 * 10-8, and 5.9 * 10-8 in that order. The electrical resistivity is generally measured at 20oC.
5. Measured using an Electrical Conductivity meter, what is the order of resistivity of superconducting materials?
a) 10-8
b) 1016
c) ∞
d) 0
View Answer
Explanation: The resistivity of superconducting materials is 0 since they conduct all electricity and resist none of it. Metals have the resistivity of the order of 10-8 while that of insulators is of the order of 1016. Since maximum electricity is refused by super-insulators, their resistivity is ∞.
6. Which of the following represents the energy band diagram of a semiconducting material?
a) 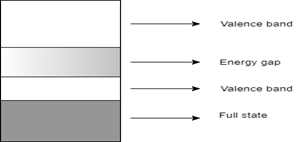
b) 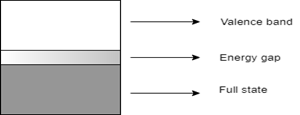
c) 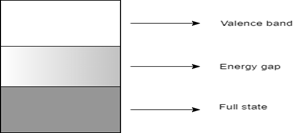
d) 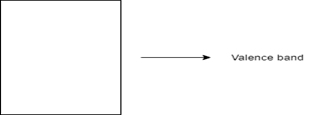
View Answer
Explanation: The diagram with the short energy gap represents the energy band diagram of semiconductor, which in this case is silicon. The diagram with multiple valence bands denotes that of a conductor, which in this case is sodium. The diagram with a wide energy gap represents an insulator like a diamond. The diagram having no energy gap is not an energy band diagram.
7. Which of the following processes is not an application of thermoelectric effect?
a) Seebeck effect
b) Peltier effect
c) Thomson effect
d) Ettingshausen effect
View Answer
Explanation: Seebeck, Peltier, and Thomson are three distinct effects which make up the thermoelectric effect. Ettingshausen effect, however, is a separate thermoelectric phenomenon like the Nernst effect.
8. The insulating capacity of material against high voltages is known as _______
a) Dielectric strength
b) Thermoelectricity
c) Electromechanical effect
d) Electrochemical effect
View Answer
Explanation: The dielectric strength of a material is its insulating capacity against a high voltage. The process by which two dissimilar metals join and produce a voltage is known as thermoelectricity. The electrical effect in a material is the relation between electrical energy and chemical change (as in batteries) and that of electromechanical effect is in an electrically operated mechanical device (as radar).
9. What is the nature of the coefficient of resistance of an insulator?
a) Positive
b) Negative
c) Zero
d) Infinite
View Answer
Explanation: The coefficient of resistance of an insulator and a semiconductor material is generally negative. It is usually positive for pure metals. Zero coefficient values can be obtained by alloying with a specific type of metals.
10. What is the dielectric strength of mica?
a) 118 MV/m
b) 2000 MV/m
c) 3 MV/m
d) 1012 MV/m
View Answer
Explanation: The dielectric strength of mica is 118 MV/m. It is generally measured at 20oC, where 1 MV/m equals 10 V/m. The dielectric strengths of diamond, air, and vacuum are 2000, and 10 in MV/m in that order.
Sanfoundry Global Education & Learning Series – Engineering Materials & Metallurgy.
To practice all areas of Engineering Materials & Metallurgy, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Apply for Metallurgical Engineering Internship
- Check Metallurgical Engineering Books
- Check Engineering Materials Books
- Practice Metallurgical Engineering MCQs
