This set of Engineering Materials & Metallurgy Multiple Choice Questions & Answers (MCQs) focuses on “Fick’s Law, Carbon Steels”.
1. What is the unit of diffusion coefficient?
a) mol m-2 s-1
b) mol m-3
c) m2 s-1
d) KJ mol-1
View Answer
Explanation: The unit of the diffusion coefficient (D) is mole per square meter per second (m2 s-1). The units of flux (J), concentration (c), and enthalpy are mol m2 s-1, mol m-3, and KJ mol-1 in that order.
2. What is Fick’s first law of diffusion?
a) ![]()
b) ![]()
c) ![]()
d) ![]()
View Answer
Explanation: Equation
3. Which of these represents concentration-distance profile for steady-state flow?
a)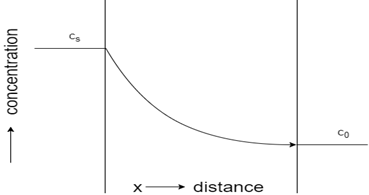
b) 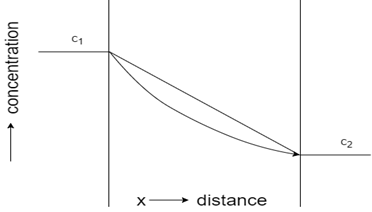
c) 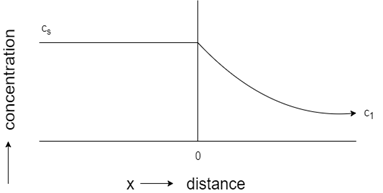
d) 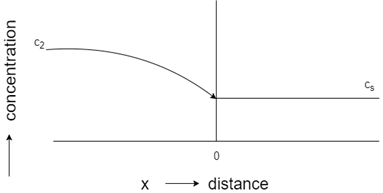
View Answer
Explanation: The profile with a straight line represents steady-state flow, whereas the curved profile indicates unsteady state flow. Initial constant concentration indicates the carburization process of steel, while constant concentration towards the end shows decarburization process of steel.
4. What is Fick’s second law of diffusion?
a) ![]()
b) ![]()
c) ![]()
d) ![]()
View Answer
Explanation: The equation
5. What is the diffusion constant (Do) when copper is dissolved in copper?
a) 0.2 * 10-4
b) 0.15 * 10-4
c) 1.98 * 10-4
d) 5400 * 10-4
View Answer
Explanation: Diffusion of Cu in Cu has a Do value of 0.2 * 10-4 m2 s-1. The diffusion constants of Zn in Zn, Al in Al, and Si in Si are 0.15 * 10-4, 1.98 * 10-4, and 5400 * 10-4 in that order.
6. Plain carbon steels are alloys mainly consisting of _________
a) Iron and Carbon
b) Potassium and Iron
c) Carbon and Potassium
d) Carbon and Indium
View Answer
Explanation: Steels are alloys having primary components as Iron and Carbon. They, however, contain elements such as silicon, manganese, phosphorous, and sulfur in varying quantities. These elements are added either intentionally, or retained during the refining process.
7. Which of the following attributes explain why pure metals are not frequently used in engineering applications?
a) Hardness
b) Brittleness
c) Softness
d) Luster
View Answer
Explanation: Pure metals are soft and ductile, which is not ideal for most engineering applications. Therefore, alloys are used to fulfill the requirements. This explains why pure gold is mixed with impurities to make ornaments.
8. Which of the following is also known as mild steel?
a) Plain carbon steel
b) Low carbon steel
c) Medium carbon steel
d) High carbon steel
View Answer
Explanation: Low carbon steels are otherwise known as mild steel owing to their carbon content. These are the carbon steels which contain less 0.30% of carbon. They are used to making small parts like wires, sheets, valves, and more.
9. What is the composition of carbon in medium carbon steels?
a) 0.05%
b) 0.1% – 0.3%
c) 0.25% – 0.6%
d) More than 0.6%
View Answer
Explanation: Medium carbon steels are those steels which contain 0.25% – 0.6% of carbon, along with traces of manganese, silicon, and copper. Dead mild contains 0.05% of carbon, whereas another mild steel contains 0.1% – 0.3% of carbon. More than 0.6% of carbon is present in high-carbon steel.
10. Which of these are applications of high-carbon steel?
a) Gears, valves
b) Steel wires, sheets, screws
c) Clutch discs, valve springs
d) Machine tools, leaf and coil springs
View Answer
Explanation: High-carbon steels with a tensile strength of 580 N/mm2 are used for making machine tools, leaf and coil springs, and similar tools. Medium-carbon steels (1230 N/mm2) are used for making clutch discs and valve springs. Mild steels (555 N/mm2) are used for making gears and valves, whereas dead mild steels (390 N/mm2) are used for making wires, sheets, and screws.
11. What is the maximum forging temperature of 1.1% carbon steel?
a) 1049oC
b) 1082oC
c) 1140oC
d) 1171oC
View Answer
Explanation: Steels with 1.1% carbon composition are forged at 1082oC, while their burning temperature is 1171oC. Steels with 1.5% carbon, however, burn at 1140oC and are forged at 1049oC.
12. Which steels are generally used for making connecting rods and gear shafts?
a) Low carbon steels
b) Medium carbon steels
c) High carbon steels
d) Stainless steels
View Answer
Explanation: Connecting rods and gear shafts are commonly made using medium carbon steels containing 0.35% to 0.45% carbon. They possess a tensile strength of 750 N/mm2. This gives the required strength and hardness to the parts.
13. What is the maximum amount of manganese in carbon steels?
a) 1.65%
b) 1.50%
c) 0.60%
d) 0.40%
View Answer
Explanation: In plain carbon steels, the composition of steel must not exceed 1.65% of manganese. It is also restricted to a maximum of 0.60% for both copper and silicon, while carbon is limited to a maximum of 1.5%.
14. What are the advantages of plain carbon steels over alloy steels?
a) Inexpensive but hard to handle
b) Expensive but easy to handle
c) Inexpensive and easy to handle
d) There are no advantages of plain carbon steels
View Answer
Explanation: Carbon steels are made of compositions having a limited quantity of elements. Due to factors such as common elements, limited quantities, and low strength, carbon steels are much cheaper than alloying steels.
Sanfoundry Global Education & Learning Series – Engineering Materials & Metallurgy.
To practice all areas of Engineering Materials & Metallurgy, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Check Metallurgical Engineering Books
- Check Engineering Materials Books
- Practice Metallurgical Engineering MCQs
- Apply for Metallurgical Engineering Internship
