This set of Corrosion Engineering Question Bank focuses on “Materials – Metals and Alloys – 2”.
1. Which of the following metals will act as sacrificial metals in cathodic production?
a) Tin and zinc
b) Zinc only
c) Magnesium, zinc and magnesium
d) Magnesium and tin
View Answer
Explanation: Sacrificial metals are the metals that corrode preferentially over the metallic structure that needs to be protected. This cathodic protection works on the principle of the galvanic effect. Tin, zinc, magnesium are the common sacrificial metals used.
2. Which of the following metal is/are corrosion resistant to hydrofluoric acid?
a) Hastelloy C
b) Monel
c) Hastelloy C and Monel
d) Titanium
View Answer
Explanation: Hastelloy C and Monel are corrosion-resistant metals to hydrofluoric acid. Whereas titanium and most other metals usually corrode in hydrofluoric acid.
3. Which of the following metal plating is used for high-strength steels in aircraft to improve resistance to corrosion fatigue?
a) Cadmium
b) Magnesium
c) Zinc
d) Lead
View Answer
Explanation: Corrosion fatigue is the simultaneous effect of fatigue and corrosion on a working metal. Cadmium plating is used for high-strength steels in aircraft to improve resistance to corrosion fatigue.
4. Which of the following metals will depend on the oxide layer for their corrosion resistance?
a) Titanium and stainless steels
b) Titanium, stainless steels, and aluminum and its alloys
c) Aluminum and its alloys
d) Aluminum and its alloy and titanium
View Answer
Explanation: Corrosion resistance of the metal is the ability of a metal to withstand the effect of a corrosive environment. Titanium, stainless steel, and aluminum and its alloys will resist the corrosive environment with the formation of their oxide layer.
5. Which of the following metals has a Hexagonal closed packed structure?
a) Cadmium
b) Magnesium
c) Zirconium
d) Cadmium, magnesium, and zirconium
View Answer
Explanation: Crystal structures are classified into 14 different types known as Bravais lattice based on the shape of the unit cell and position of atoms in it. Cadmium, magnesium, zirconium, titanium, zinc, and beryllium are examples of hexagonal closed packed structures.
6. Which of the following characteristics of titanium that account for much of its application in corrosive services?
a) Resistance to seawater and other chloride salt solutions
b) Resistance to hypochlorite’s and wet chlorine
c) Resistance to fuming nitric acid
d) Resistance to seawater, chloride salt solutions, hypochlorite’s, wet chlorine and fuming nitric acid
View Answer
Explanation: Titanium is an excellent corrosion-resistant metal in many corrosive environments. The characteristics of titanium that account for much of its application in corrosion services are its resistance to seawater, chloride salt solutions, hypochlorite’s, wet chlorine, and fuming nitric acid.
7. Which of the following metals are regarded as refractory metals?
a) Niobium and tungsten
b) Molybdenum only
c) Niobium, molybdenum, and tungsten
d) Tungsten and molybdenum only
View Answer
Explanation: Refractory metals are characterized by very high melting points with limited corrosion resistance. Tantalum, niobium, molybdenum, tungsten, and zirconium are regarded as refractory metals.
8. Which of the following metals that usually results in a porous and volatile oxide layer?
a) Magnesium
b) Molybdenum
c) Sodium
d) Magnesium, sodium, and molybdenum
View Answer
Explanation: If the Pilling-bed worth ratio of a particular metal is less than 1 then it results in a porous, volatile, and thin oxide layer. Sodium, molybdenum, and magnesium are examples of this kind.
9. Which of the following metals are regarded as noble metals?
a) Iridium
b) Palladium
c) Ruthenium
d) Iridium, palladium, and ruthenium
View Answer
Explanation: Noble metals are the metals that are characterized by highly positive potentials relative to the hydrogen electrode along with excellent corrosion resistance. This includes iridium, palladium, ruthenium, rhodium, osmium, gold, silver, and platinum.
10. What is the composition of aqua-regia that corrodes noble metals such as platinum, gold?
a) 3 parts of nitric acid and 1 part of hydrochloric acid
b) 3 parts of hydrochloric acid and 1 part of nitric acid
c) 1 part of hydrofluoric acid and 3 part of hydrochloric acid
d) 3 parts of hydrofluoric acid and 1 part of hydrochloric acid
View Answer
Explanation: The composition of aqua regia is 3 parts of hydrochloric acid and 1 part of nitric acid. It corrodes/dissolves noble metals such as gold, platinum, and palladium.
11. What is the abbreviation of BMG regarding the type of materials?
a) Bulk metallic groups
b) Bulk metallic glasses
c) Body metallic glasses
d) Body metallic groups
View Answer
Explanation: Bulk metallic glasses are the non-crystalline alloys that are formed with low critical cooling rates. These bulk metallic glasses exhibit useful engineering properties such as high mechanical strength and high corrosion resistance.
12. Metallic composites consist of a metallic matrix strengthened by metallic or nonmetallic fibers, filaments or whiskers.
a) True
b) False
View Answer
Explanation: Composites are the type of materials that are formed by the combination of two or more materials. And metallic composites consist of a metallic matrix strengthened by metallic or nonmetallic fibers or whiskers. This includes boron, graphite, glass, and metal.
13. Which of the following factors that affect the corrosion of composites?
a) The corrosive specificity of an environment
b) Galvanic effect
c) Corrosive specificity of an environment and galvanic effect
d) Neither the corrosive specificity of an environment nor the galvanic effect
View Answer
Explanation: The corrosion specificity of an environment and galvanic effect are the factors that affect the corrosion of composites. Thus, if the matrix and filament of a composite are close together in the galvanic series. galvanic effects are negligible.
14. Which of the following is the monomer of natural rubber?
a) Isoprene
b) Neoprene
c) Vinyl chloride
d) Styrene
View Answer
Explanation: Natural rubber is a long-chain molecule of isoprene which is chemically known as polyisoprene. It is obtained from trees as liquid latex. The properties of natural rubber can be improved by the process known as vulcanization of rubber.
15. What is the packing fraction of a crystal structure that is depicted in the given figure?
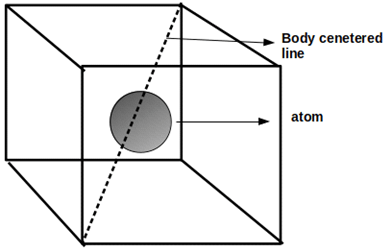
a) 0.74
b) 0.52
c) 0.68
d) 0.77
View Answer
Explanation: The packing fraction of a given crystal structure is defined as the ratio of the volume of atoms in the unit cell to the volume of the unit cell. Given crystal structure is body-centered cubic and its packing fraction is 0.68.
Sanfoundry Global Education & Learning Series – Corrosion Engineering.
To practice Corrosion Engineering Question Bank, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Apply for Mechanical Engineering Internship
- Apply for Metallurgical Engineering Internship
- Practice Metallurgical Engineering MCQs
- Practice Mechanical Engineering MCQs
- Check Corrosion Engineering Books
