This set of Refrigeration Multiple Choice Questions & Answers (MCQs) focuses on “Air Refrigerator Working on Reverse Carnot Cycle – 1”.
1. For a refrigerating system that works on reversed Carnot cycle having vapor as refrigerant, which one of the following is not a process of the cycle in p-v diagram?
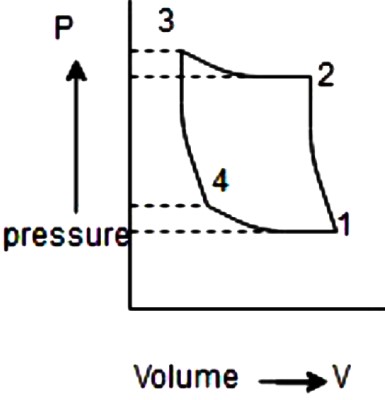
a) Isentropic compression process
b) Isothermal compression process
c) Adiabatic expansion process
d) Isothermal expansion process
View Answer
Explanation: The reversed Carnot cycle has 4 processes namely, isothermal compression process, isentropic compression process, isentropic expansion process and isothermal expansion process.
2. What is the coefficient of performance of the refrigerant system working on reversed Carnot cycle, having Qa as heat absorbed by the air during isothermal expansion per kg of air and Qr as heat rejected during isothermal compression per kg of air?
a) COP=\(\frac{Q_a}{(Q_r – Q_a)}\)
b) COP=\(\frac{Q_r}{(Q_r – Q_a)}\)
c) COP=\(\frac{Q_a}{(Q_r + Q_a)}\)
d) COP=\(\frac{Q_a}{(Q_a – Q_r)}\)
View Answer
Explanation: COP of refrigerator is COP of heat pump – 1
i.e. \([\frac{Q_r}{Q_r – Q_a}] – 1 = \frac{Q_a}{(Q_r – Q_a)}\).
3. The COP of refrigerator may be improved by _________
a) decreasing the higher (T2) as well as the lower (T1) temperature
b) decreasing the higher (T2) temperature and increasing the lower (T1) temperature
c) increasing the higher (T2) as well as the lower (T1) temperature
d) keeping the higher (T2) temperature the same while decreasing the lower (T1) temperature
View Answer
Explanation: The COP of refrigerator can be given by :
COP = \(\frac{T_1}{(T_2- T_1)}\). Hence by decreasing the higher temperature T2 or by increasing the lower temperature T1 the COP may be improved.
4. What happens to the COP of a Carnot refrigerator in summer and in winter?
a) The COP is more in winter
b) The COP is more in summer
c) The COP remains unaffected
d) The COP fluctuates continuously during winter and summer
View Answer
Explanation: As the higher temperature T2 will always be lower in winter when compared with that of summer i.e. the temperature of air available of heat rejection will be low, the COP in winter will be higher.
5. A refrigerating machine working on Carnot cycle operates between 300K and 250K. Determine the COP.
a) 5
b) 6
c) 4
d) 10
View Answer
Explanation: The COP of refrigerating machine is given by
COP = \(\frac{T_1}{(T_2- T_1)} = \frac{250}{(300-250)}\)=5.
6. The COPr of a Carnot refrigerating machine is 7.89. What will be the COPp of heat pump?
a) 10.3
b) 7.89
c) 8.89
d) 6.89
View Answer
Explanation: Relation between COP of heat pump and COP of the refrigerating machine is
COPr = COPp-1
7.89 = COPp-1
COPp=7.89+1=8.89.
7. Which one is not a limitation of the Carnot cycle with air or gas as refrigerant?
a) It has low efficiency when working between two fixed temperature limits
b) Machine has to run at high and low speeds during adiabatic and isothermal process. Such variation of speed is not possible
c) Extreme pressure and large volume are developed
d) It is not possible to carry out isothermal heat transfer process in practice
View Answer
Explanation: Carnot cycle has the highest efficiency theoretically when working between two fixed temperature limits. Other limitations of Carnot cycle are machine has to run at high and low speeds during adiabatic and isothermal process. Such variation of speed is not possible, extreme pressure and large volume is developed, it is not possible to carry out isothermal heat transfer process in practice.
8. For a refrigerating system that works on Carnot cycle having air or gas as refrigerant s, which one of the following is not a process of the cycle in p-v diagram?

a) Adiabatic compression process
b) Isothermal compression process
c) Adiabatic expansion process
d) Isentropic expansion process
View Answer
Explanation: The Carnot cycle having air or gas as refrigerant has 4 processes namely isothermal compression process, adiabatic compression process, isothermal expansion process and adiabatic expansion process.
9. What is the COP of a refrigerator when it’s refrigerating effect R is 6.2755 KW and work consumed is 0.82 KW?
a) 8.8
b) 6.77
c) 7.65
d) 8.56
View Answer
Explanation: COP of refrigerator = Refrigeration effect / work consumed
COP=\(\frac{R}{wc}=\frac{6.2755}{0.82}\)=7.65.
10. Out of the following reasons which one is responsible for Carnot cycle not being used in practice?
a) It gives low COP
b) It gives high refrigeration effect
c) It gives low refrigeration effect
d) It has low theoretical efficiency
View Answer
Explanation: Carnot cycle is not used in everyday practice and one of the many reasons is that because it has low refrigeration effect, even though it has the highest theoretical efficiency and also it is not possible to carry out isothermal heat transfer in practice.
Sanfoundry Global Education & Learning Series – Refrigeration & Air Conditioning.
To practice all areas of Refrigeration & Air Conditioning, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Check Refrigeration & Air Conditioning Books
- Practice Mechanical Engineering MCQs
- Check Mechanical Engineering Books
- Apply for Mechanical Engineering Internship
