This set of Protein Engineering Multiple Choice Questions & Answers (MCQs) focuses on “Structure Function Relationships – Leucine Zippers”.
1. Which of the following motif contains an amphipathic alpha-helix with a series of hydrophobic amino acid residues on one side?
a) Homeodomain
b) Zinc fingers
c) H-T-H motif
d) Leucine zipper
View Answer
Explanation: Leucine zipper motif contains an amphipathic alpha-helix with a series of hydrophobic amino acid residues on one side. It is made up of two alpha-helices. Hydrophobic surface forms an area of contact between the two polypeptides of a dimer.
2. Leucine residues in the leucine zipper do not interdigitate.
a) False
b) True
View Answer
Explanation: The above statement is true. Leucine residues in the leucine zipper do not interdigitate. Although it was initially thought that leucine residues in the leucine zipper interdigitate, but we now know that they line up side by side.
3. Which of the following motifs form a coiled-coil?
a) Zinc fingers
b) Homeodomain
c) H-T-H motif
d) Leucine zipper
View Answer
Explanation: Leucine zippers form a coiled-coil structure. The leucine residues line up side by side as the interacting alpha-helices coil around each other and thus form a coiled-coil. Zinc fingers, homeodomain, and H-T-H motif do not form a coiled-coil structure.
4. The DNA-binding domain of regulatory proteins (having leucine zippers) are rich in which amino acid residues?
a) Met or Ala
b) Gly or Trp
c) His or Cys
d) Lys or Arg
View Answer
Explanation: The DNA-binding domain of regulatory proteins (having leucine zippers) are rich in Lys or Arg amino acid residues. Lys and Arg are basic amino acids that can interact with the negatively charged phosphates of the DNA.
5. In the zipper region of leucine zippers, what is the occurrence of leucine residues?
a) At every 9th position
b) At every 11th position
c) At every 4th position
d) At every 7th position
View Answer
Explanation: In the zipper region of leucine zippers, leucine occurs at every 7th position. These leucine residues have a role in the lining up of the two alpha-helices side by side. The hydrophobic interactions between these residues stabilize this interaction.
6. The two alpha-helices of the leucine zipper are present on different subunits of a dimeric regulatory protein.
a) False
b) True
View Answer
Explanation: The above statement is true. The two alpha-helices of the leucine zipper are present on different subunits of a dimeric regulatory protein. These two alpha-helices are zipped together upon DNA binding, hence, called as coiled-coil.
7. In leucine zippers, the DNA-binding region and the protein-binding region are the same.
a) True
b) False
View Answer
Explanation: The above statement is false. In leucine zippers, the DNA-binding region and the protein-binding region are not the same. They are present on different sides of the two alpha-helices. The two alpha-helices wrap around each other in a gently coiled-coil.
8. Which of the following DNA-binding motif is shown in the figure below?
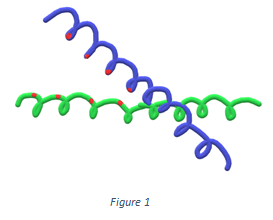
a) Homeodomain
b) Zinc fingers
c) H-T-H motif
d) Leucine zipper
View Answer
Explanation: The DNA-binding motif which is shown in the figure above is the leucine zipper. It consists of two alpha-helices from two different subunits of a protein, wrapped around in a coiled-coil fashion.
9. The amino acid leucine is critical for the functioning of which motifs?
a) H-T-H motif
b) Homeodomain
c) Zinc fingers
d) Leucine zipper
View Answer
Explanation: The amino acid leucine is critical for the functioning of the leucine zipper DNA-binding motif. Leucine occurs at every seventh position in the protein interacting region of the leucine zipper.
10. The B-ZIP (basic-region leucine zipper) class of eukaryotic transcription factors contain which of the following DNA-binding motif?
a) Homeodomain
b) Zinc fingers
c) H-T-H motif
d) Leucine zipper
View Answer
Explanation: The B-ZIP (basic-region leucine zipper) class of eukaryotic transcription factors contain a leucine zipper DNA-binding motif. It does not contain homeodomain or zinc fingers or an H-T-H motif.
11. The leucine zipper is a left-handed parallel dimeric coiled-coil.
a) False
b) True
View Answer
Explanation: The above statement is true. The leucine zipper is a left-handed parallel dimeric coiled-coil. It is named so because it contains leucine amino acid residues on one side of alpha-helices. The C terminus is an amphipathic alpha-helix that dimerizes to form the leucine zipper.
12. Which of the following region of the alpha-helix in leucine zippers is amphipathic?
a) Either C or N terminus
b) Both C and N terminus
c) N terminus
d) C terminus
View Answer
Explanation: The C terminus region of the alpha-helix in leucine zippers is amphipathic. It dimerizes to form a leucine zipper. An amphipathic alpha-helix has two surfaces along its length, one side is hydrophilic and the other side is hydrophobic.
13. Which of the following DNA-binding motif is also known as leucine scissors?
a) Homeodomain
b) H-T-H motif
c) Zinc fingers
d) Leucine zipper
View Answer
Explanation: Leucine zipper is the DNA-binding motif that is also known as leucine scissors. They are called leucine scissors because they look like scissors. This scissor-like structure is formed by the two alpha-helices crossing over each other.
14. Leucine zippers are present only in eukaryotic regulatory proteins?
a) True
b) False
View Answer
Explanation: The above statement is false. Leucine zippers are not only present in eukaryotic regulatory proteins, but they are present in prokaryotic regulatory proteins also. Although they are mainly a feature of eukaryotes.
15. The basic residues in the DNA-binding domain of leucine zippers interact with which of the following position in the DNA?
a) Minor groove and sequence-independent manner
b) Major groove and sequence-independent manner
c) Minor groove and sequence-dependent manner
d) Major groove and sequence-dependent manner
View Answer
Explanation: The basic residues in the DNA-binding domain of leucine zippers interact with the major groove of the DNA in a sequence-dependent manner. They do not interact in the minor groove of the DNA and neither they interact in a sequence-independent manner.
Sanfoundry Global Education & Learning Series – Protein Engineering.
To practice all areas of Protein Engineering, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Apply for Biotechnology Internship
- Check Biotechnology Books
- Practice Biotechnology MCQs
- Check Protein Engineering Books
