This set of Solid State Chemistry Questions and Answers for Experienced people focuses on “Differential Thermal Analysis and Differential Scanning Calorimetry”.
1. During DTA (Differential thermal analysis) what kind of reference material is used?
a) Chemically active
b) Physically active
c) Inert
d) Having catalytic property
View Answer
Explanation: Differential thermal analysis is a technique in which the temperature sample is compared with that of an inert material during a programmed change of temperature.
2. In DTA, the ___________________ of sample and reference should be______________ until any thermal event take place. Fill up the appropriate options for the two blanks given from the choices given below.
a) Concentration, different
b) Concentration, same
c) Temperature, different
d) Temperature, same
View Answer
Explanation: In the differential thermal analysis, DTA method, the temperature of the sample and reference material should always be same until any thermal event takes place such as melting, decomposition or cage in crystal structure, occurs in the sample, in which case the temperature either lags behind (if endothermic) or leads (if exothermic) the reference temperature.
3. What is the factor that limits the application of this differential thermal analysis method?
a) Huge apparatus
b) Insensitivity
c) Highly reactive
d) Low growth rate
View Answer
Explanation: Because of the insensitivity of the DTA differential thermal analysis method, this technique has very limited applications, its main use has been in the method of cooling curves which was used to determine the phase diagram.
4. In DTA method, the sample temperature was recorded on __________
a) Heating
b) Cooling
c) Gaseous
d) Liquid
View Answer
Explanation: In the differential thermal analysis the sample temperature I recorded on cooling rather than on heating and since the heat effects associated with solidification and crystallization are usually large, they could be detected by this method.
5. What is the reading of output when the sample and reference are at same temperature in DTA?
a) Negligible
b) Zero
c) Very high
d) Moderate
View Answer
Explanation: When the sample and the reference material are at the same temperature, the ∆net output of this pair of thermocouples is zero. When a thermal event occurs in the sample, a temperature difference, ∆T, exists between the sample and reference which is detected by the net voltage of the thermocouples.
6. What kind of material is used to monitor the temperature of heating in a differential thermal analysis?
a) Active electrolyte
b) Inactive electrolyte
c) An electrode
d) Thermocouple
View Answer
Explanation: In DTA method (differential thermal analysis) a third thermocouple is used to monitor the temperature of the heating block and the results are presented as ∆T against temperature.
7. In the normal DTA instruments, what is the size of the sample size to be used?
a) Very large
b) Moderate
c) Small
d) Very very small
View Answer
Explanation: Commercial DTA instruments are available which enable the temperature range-190 to 1600 degree Celsius to be covered. Sample sizes are usually small, a few milligrams because then there is less trouble with thermal gradients within the sample which could lead to reduced sensitivity and accuracy.
8. What is the main factor because of which DTA cells are designed?
a) Maximum sensitivity
b) Minimum sensitivity
c) Maximum solubility
d) Minimum solubility
View Answer
Explanation: DTA, differential thermal analysis cells are usually designed for maximum sensitivity to thermal changes but this is often at the expense of losing a calorimetric response.
9. What is the main factor that differentiates DTA cells and DSA cell?
a) Sensitivity
b) Thermal conductivity
c) Nature of the cell
d) Designing of the cell
View Answer
Explanation: DSC and DTA cells are very similar. A sample and an inert reference are also used in DSC but the cell is designed differently. In some DSC cells, the sample and reference are maintained at the same temperature during the heating programme and the extra heat input to the sample required in order to maintain this balance is measured.
10. Which of the following method is described by the diagram given below?
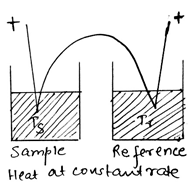
a) Hydrothermal analysis
b) Thermogravitric analysis
c) Heat analysis
d) Differential thermal analysis
View Answer
Explanation: The arrangement normally used in DTA, differential thermal analysis. Sample and reference are placed side by side in a heating block which is either heated or cooled at a constant rate, identical thermocouples are placed in each and are connected ‘back to back’.
Sanfoundry Global Education & Learning Series – Solid State Chemistry.
To practice all areas of Solid State Chemistry for Experienced people, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Check Solid State Chemistry Books
- Practice Biotechnology MCQs
- Apply for Biotechnology Internship
- Check Biotechnology Books
