This set of Pulp and Paper Multiple Choice Questions and Answers (MCQs) focuses on “Wet and Dry Strength”.
1. Wet strength additives work by forming ionic bonds between fibers.
a) True
b) False
View Answer
Explanation: The system works by forming their own cross-linked network of covalent bonds. It is a type of bonding where pair of electrons are shared. These electron pairs are called as shared pairs or bonding pairs, and the stable balance of attractive and repulsive forces between atoms.
2. It is well known that _________ cause maximum flocculation shortly after their addition to fiber slurries.
a) Anionic polymer
b) Cationic polymer
c) Lyophobic
d) Lyophilic
View Answer
Explanation: This was observed by Koethe and Scott in 1993.The time on the order of 10 to 100 or more minutes zeta potential decreases.
3. _________ are too large to easily be measure by their zeta potential.
a) Filbers
b) Cationic polymer
c) Lyophobic
d) Lyophilic
View Answer
Explanation: Fibers are too large to easily measure their zeta potential. Often the assumption is made that the zeta potential behaviour of fiber fines is the same as that of the fibers.
4. What’s the name of the group?
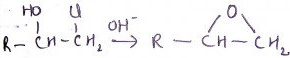
a) Carbonate
b) Epoxy
c) Polymer
d) Starch
View Answer
Explanation: The epoxy groups of this and other molecules that use it (such as materials utilised to prepare cationic starch) are stored in a stable form and made reactive before using them.
5. The _________ can be measured by the movement of electrophoresis.
a) Zeta resistance
b) Voltage
c) Current
d) Zeta potential
View Answer
Explanation: The electric field cautilises movement of the water, so the measurements must be made where it is stable . A microscope must be utilised to view the particles and determine their velocity to briefly study zeta potential.
6. What is the name of the hidden part?
_________= EZ/f
a) Voltage
b) Zeta potential
c) Current
d) Factor
View Answer
Explanation: The velocity of migration of a molecule is proportional to the electric field (E), which is the volts of the field that’s divided by the distance b/w the plates, also referred to the voltage drop per unit length, V/m and the charge of the species (Z) and is indirectly proportional to the frictional coefficient, (f).
7. What’s the name of the instrument?
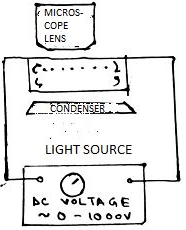
a) Electrocuting
b) Electrophoresis
c) Electrolysis
d) Electro-magnetism
View Answer
Explanation: Electrophoresis is a technique utilized in laboratories in order to separate macromolecules based on size. The technique applies a -ve charge so proteins move towards a +ve charge.
8. Colloidal _________ have a disadvantage that it’s hard to reproduce the method from operator to operator without mechanization of the system.
a) Titration
b) Sulfonication
c) Burning
d) Delignification
View Answer
Explanation: The endpoint determination isn’t very sharp to the unaided eye. The use of a spectroscopic instrument to help determine the endpoint is crucial but adds to the complexity and cost of the method.
9. In the direct titration methods, the indicator isn’t added to the solution containing the cationic polymer.
a) True
b) False
View Answer
Explanation: Anionic polymer is added by a burett. Initially it complexes completely with the cationic polymer.
10. Problems in zeta potential measurement occur if the H2O has a relatively high _________strength since heating will occur, which induces convection currents.
a) ionic
b) magnetising
c) acid
d) basic
View Answer
Explanation: Decrement the applied voltage or measurement time will be necessary. Samples must be dilute with a solution of similar inorganic electrolyte composition to observe individual particles.
Sanfoundry Global Education & Learning Series – Pulp and Paper.
To practice all areas of Pulp and Paper, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Check Chemical Engineering Books
- Practice Chemical Engineering MCQs
- Apply for Chemical Engineering Internship
