This set of Class 11 Physics Chapter 11 Multiple Choice Questions & Answers (MCQs) focuses on “Thermal Properties of Matter – Change of State”.
1. Change of phase occurs at a constant temperature, which implies that even on addition of heat to a substance temperature remains constant. True or False?
a) True
b) False
View Answer
Explanation: The temperature change with heat supplied is shown below. During change of phase heat supplied is used to make changes in intermolecular distances while the temperature remains constant.
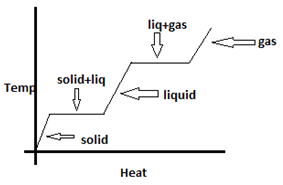
2. Melting point depends on pressure. True or False?
a) True
b) False
View Answer
Explanation: Melting point is less for higher pressure. For eg: we can keep a small weight of 1kg on a block of ice and we will see that the ice underneath the block starts melting at a lower temperature.
3. What is regelation?
a) Refreezing of ice on addition of impurities
b) Refreezing of ice on reduction in pressure
c) Melting of ice at lower temperature than its melting point due to increase in pressure
d) Melting of ice at higher temperature than its melting point
View Answer
Explanation: When increase in pressure causes water to melt. It may refreeze on removal of this extra pressure. This refreezing is called relegation.
4. What happens to the boiling point of water in a container when the container is completely closed by a lid?
a) Increases
b) Decreases
c) Remains same
d) Boiling point is characteristic of a substance
View Answer
Explanation: Boiling occurs when vapour pressure of liquid is the same as atmospheric pressure. The vapour pressure increases with increase in temperature. So when temperature has been increased sufficiently boiling starts. But with increase in pressure at higher vapour pressure has to be reached which is obtained by increasing temperature and therefore we say boiling point increases.
5. Select the correct statement
a) On heating continuously a substance changes from solid to gas via liquid phase only
b) Change of phase occurs at variable temperature
c) Water can exist in all 3 phases
d) Boiling point increases with increase in pressure
View Answer
Explanation: Water can exist in all 3 phases that are in thermal equilibrium with each other. It is referred to as the triple point of water. When a substance is heated it may change directly from solid to gas. This is called sublimation. Boiling point increases with decrease in pressure. Change of phase occurs at constant pressure.
6. Getting burnt by steam is less dangerous than boiling water. True or False?
a) True
b) False
View Answer
Explanation: To make steam, water has to be boiled and then more heat energy has to be added to convert it into steam. So, steam is more dangerous than boiling water.
7. If 5g of a solid requires 30cal of heat to change into liquid at the same temperature, calculate the latent heat of fusion in cal/g.
a) 150
b) 0.167
c) 6
d) 3
View Answer
Explanation: For changing a solid into liquid, heat has to be supplied and this change occurs at constant temperature. Q = mL
⇒ 30 = 5L
⇒ L = 6 cal/g,
8. The diagram given below shows phase changes for a substance based on experimental observations. Select the correct statement.
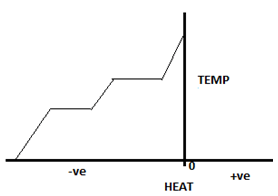
a) Heat is being removed from body while increasing temperature
b) Heat is constant during phase change
c) Temperature is in Kelvin
d) The two horizontal lines in the graph represent phase change as the temperature is constant
View Answer
Explanation: The diagram represents phase change from solid to liquid to gas. Heat removal leads to decrease in temperature. The temperature becomes zero at an instant, so it can’t be in Kelvin as 0K has never been achieved. The two horizontal lines represent phase change as temperature remains constant at that time.
9. Condensation temperature is the same as evaporation temperature. True or False?
a) True
b) False
View Answer
Explanation: Condensation is the conversion of vapour to liquid, while evaporation is the conversion of liquid to vapour. Both occur at the same temperature, the only difference being that heat is removed in the former and added in the latter. The graph as shown below is a straight line at constant temperature.
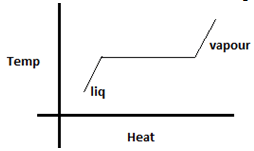
10. Select the correct statement for melting of any substance.
a) It occurs at the same temperature as that of vapourization
b) It is exothermic in nature
c) It occurs at 100°C
d) It requires heat to be supplied to the substance
View Answer
Explanation: Melting is the conversion of solid to liquid at constant temperature. It is endothermic and the melting temperature varies from substance to substance and also on atmospheric conditions like pressure.
Sanfoundry Global Education & Learning Series – Physics – Class 11.
To practice all chapters and topics of class 11 Physics, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Practice Class 11 - Chemistry MCQs
- Check Class 11 - Physics Books
- Practice Class 11 - Mathematics MCQs
- Practice Class 11 - Biology MCQs
- Practice Class 12 - Physics MCQs
