This set of Materials Science Mcqs focuses on “Binary Phase Diagrams”.
1. How many components are present in binary phase system?
a) 3
b) 4
c) 2
d) 1
View Answer
Explanation: In binary phase system there will be only two components.
2. The line above which the alloy is liquid is called __________
a) Solidus line
b) Tie line
c) Liquidus line
d) Lever line
View Answer
Explanation: The liquidus line is the line above which the alloy is liquid. At the temperature just below this line crystal of solid solution start forming.
3. It can be noted that the two substances are soluble in each other in the entire range of compositions in both liquid and solid state. This kind of system is known as ___________
a) Binary phase system
b) Unary phase system
c) Multiple phase system
d) Isomorphous system
View Answer
Explanation: Isomorphous phase diagram is the phase diagram in which the two components are soluble in each other over an entire range of composition in both solid and liquid phases.
4. What is the XY line in the given phase diagram?
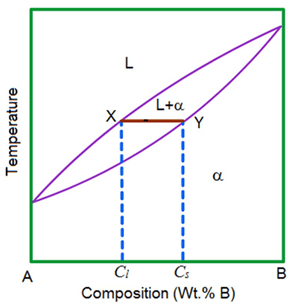
a) Lever line
b) Tie line
c) Solidus line
d) Liquidus line
View Answer
Explanation: To find the composition of the individual phases in the two phase region, a line (XY) is drawn and its intercepts on the liquidus and solidus lines. This is called as tie line.
5. What is the α region in the given phase diagram?

a) Homogeneous liquid solution
b) Semi solid solution
c) Vapor
d) Substitutional solid solution
View Answer
Explanation: α-region is a substitutional solid solution of two components A and B. And the given phase diagram denotes the Cu-Ni phase diagram.
6. What is used for the determination of Phase amounts?
a) Tie line and Temperature–Composition Point
b) Lever line
c) Temperature–Composition Point
d) Pressure
View Answer
Explanation: Lever rule is used to determine the relative amount of each phase in a mixture. It takes the help of a tie-line at a given temperature.
7. A 53% Ni Cu-Ni alloy is cooled from the liquid state to 1300°C. Calculate the % of liquid and solid at 1300°C.
a) 28, 72
b) 38, 62
c) 35, 65
d) 65, 35
View Answer
Explanation: The tie line at 1300ﹾC intersects solidus at 58% Ni and liquidus at 45% Ni.
Apply the lever rule to get the liquid fraction
% Liquid = 100* (5)/(13) = 38%
%Solid = 100* (53 – 45)/(58 – 45) = 62% (100 – %Liquid).
8. According to the given figure, which of the following is used to determine the amount of each phase in a mixture?
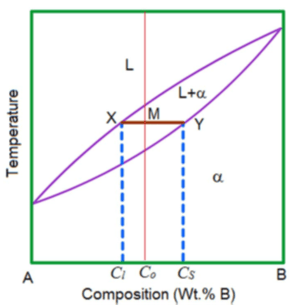
a) Lever rule
b) Tie line
c) Solidus line
d) Liquidus line
View Answer
Explanation: For two phase system lever rule helps i.e. the tie line must be utilized in conjunction with a procedure that is often called the lever rule.
9. What is the equation for liquid mass fraction of this given phase diagram?

a) cα – c0 / cα – cL
b) c0 – cL / cα – cL
c) cα / cα – cL
d) c0 / cα – cL
View Answer
Explanation: Cα is composition of solid, CL is composition of liquid, C0 is composition of alloy.
10. What is the region between liquidus and solidus lines?

a) Two-phase region where liquid and solid coexist
b) Solid region
c) Liquid region
d) Lava region
View Answer
Explanation: The intermediate region between liquidus and solidus lines is the two-phase region where liquid and solid coexists.
Sanfoundry Global Education & Learning Series – Materials Science.
To practice MCQs on all areas of Materials Science, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
