This set of Mass Transfer Multiple Choice Questions & Answers (MCQs) focuses on “Concentrations, Velocities And Fluxes”.
1. The ratio of a mass concentration of species A to the total mass density of the mixture is known as
a) Mass density
b) Concentration
c) Mole fraction
d) Mass fraction
View Answer
Explanation: It is defined as p a/p.
2. For a binary mixture of species A and B, the mass average velocity is defined as
a) 2 pa Va + pb Vb/ p
b) pa Va + pb Vb/ p
c) pa Va + 2 pb Vb/ p
d) pa Va + pb Vb/ 2 p
View Answer
Explanation: pa Va + pb Vb/ pa + pb = pa Va + pb Vb/ p.
3. The ratio of number of moles of species A to the total number of moles of the mixture is known as
a) Mole fraction
b) Mass fraction
c) Partial pressure
d) Mass density
View Answer
Explanation: It is defined as na/n.
4. A binary mixture of oxygen and nitrogen with partial pressures in the ratio 0.21 and 0.79 is contained in a vessel at 300 K. If the total pressure of the mixture is 1 * 10 5 N/m2, find molar concentration of oxygen
a) 8.42 * 10-1 kg mol/m3
b) 8.42 * 10-2 kg mol/m3
c) 8.42 * 10-3 kg mol/m3
d) 8.42 * 10-4 kg mol/m3
View Answer
Explanation: 0.21 * 10 5/8341 * 300 = 8.42 * 10 -3 kg mol/m3.
5. For a binary mixture of species A and B, the molar average velocity is defined as
a) n a V a + n b V b
b) n a V a + n b V b/n
c) n a V a + n b V b/2 n
d) n a V a + 2 n b V b/n
View Answer
Explanation: n A V A + n B V B/n A + n B = n A V A + n B V B/n.
6. Given diagram shows diffusion of water vapor through air. Identify the correct statement
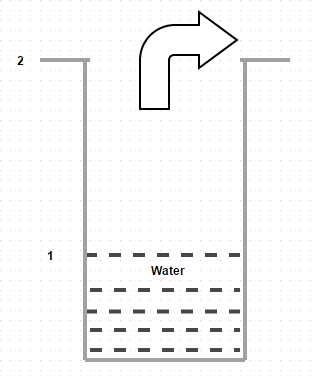
a) The water evaporates and diffuses downward
b) The water does not evaporates but diffuses upward
c) The water evaporates and diffuses upward
d) The water does not evaporates but diffuses downward
View Answer
Explanation: The slight movement of air over the top of the tube does not bring about any change in the concentration profile of air.
7. A binary mixture of oxygen and nitrogen with partial pressures in the ratio 0.21 and 0.79 is contained in a vessel at 300 K. If the total pressure of the mixture is 1 * 10 5 N/m2, find overall mass density
a) 4.156 kg/m3
b) 3.156 kg/m3
c) 2.156 kg/m3
d) 1.156 kg/m3
View Answer
Explanation: For oxygen = 0.269 kg/m3 and for nitrogen = 0.887 kg/m3.
8. A binary mixture of oxygen and nitrogen with partial pressures in the ratio 0.21 and 0.79 is contained in a vessel at 300 K. If the total pressure of the mixture is 1 * 10 5 N/m2, find molar fraction of nitrogen
a) 0.79
b) 0.21
c) 0.23
d) 0.13
View Answer
Explanation: Molar fraction is equal to the partial pressure.
9. A binary mixture of oxygen and nitrogen with partial pressures in the ratio 0.21 and 0.79 is contained in a vessel at 300 K. If the total pressure of the mixture is 1 * 10 5 N/m2, find mass fraction of oxygen
a) 0.133
b) 0.233
c) 0.333
d) 0.433
View Answer
Explanation: 0.269/1.156 = 0.233.
10. A binary mixture of oxygen and nitrogen with partial pressures in the ratio 0.21 and 0.79 is contained in a vessel at 300 K. If the total pressure of the mixture is 1 * 10 5 N/m2, find the average molecular weight of the mixture
a) 28.84
b) 29.84
c) 30.84
d) 31.84
View Answer
Explanation: (0.21) (32) + (0.79) (28) = 28.84.
Sanfoundry Global Education & Learning Series – Mass Transfer.
To practice all areas of Mass Transfer, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Check Mass Transfer Books
- Apply for Mechanical Engineering Internship
- Practice Chemical Engineering MCQs
- Apply for Chemical Engineering Internship
- Check Chemical Engineering Books
