This set of Finite Element Method Question Bank focuses on “Single Variable Problems – Applications – 2”.
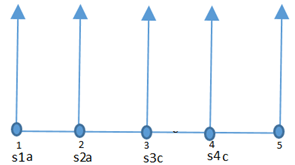
1. A river flows along node 1 through node 5, infiltrating an aquifer at a constant rate. What is the value of s1a+s2a+s3c+s4c if s denotes a linear interpolation function in FEM?
a) 1
b) 2
c) 3
d) 4
View Answer
Explanation: Since s denotes a linear interpolation function in FEM, the sum of the two linear interpolation functions corresponding to one element must be equal to unity. Thus, s1a+s2a =1 and s3c+s4c=1.
→s1a+s2a+s3c+s4c =2.
2. In a thermodynamics process, what is the correct term for the series of states through which a system passes?
a) Path
b) Phase
c) Cycle
d) Direction
View Answer
Explanation: A thermodynamic path is series of states through which a system passes from an initial state of equilibrium to a final state of equilibrium and can be plotted on a pressure-volume (P-V), pressure-temperature (P-T), and temperature-entropy (T-s) diagrams to view graphically. Phase is a quantity of matter that can be separated mechanically from a heterogeneous mixture.
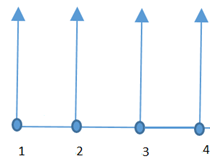
3. A river flows along node 1 through node 4, infiltrating an aquifer at constant rate of 5(m3/day*m2). The length of elements shown is 2m. What is the value of global force due to infiltration of the river at node 2?
a) 5
b) 10
c) 15
d) 20
View Answer
Explanation: The value of global force due to infiltration of the river at node 2 is given by 0.5*q*h+0.5*q*h, where 0.5 denotes the value of the two shape functions, q is rate of infiltration and h is length of the element.
=0.5*q*h+0.5*q*h
=q*h
Given q=5, h=2
=5*2
=10.
4. In which thermodynamics process the temperature remains constant?
a) Isobaric
b) Isentropic
c) Isothermal
d) Isochoric
View Answer
Explanation: An Isobaric process takes place at constant pressure an Isentropic process takes place at constant entropy. An Isothermal process takes place at constant temperature whereas an isochoric process takes place at constant volume.
5. If ψ=2x+3y represents stream function then what is the magnitude of velocity?
a) 3
b) 5
c) 6.3
d) 3.6
View Answer
Explanation: The magnitude of velocity (v) is (V2+U2)(1/2).
V=\(\frac{\partial \psi}{\partial y}\)=3 and U=\(\frac{-\partial \psi}{\partial x}\)=-2
v=(32+22)(1/2)
v=(9+4)(1/2)
=3.6.
6. In which process does a thermodynamic system remains infinitesimally closed to an equilibrium state at all times?
a) Path equilibrium process
b) Cycle equilibrium process
c) Phase equilibrium process
d) Quasi-state or quasi-equilibrium process
View Answer
Explanation: Quasi-static Process, in thermodynamics, is a process that happens infinitely slowly. However, it is very important to note that no real process is Quasistatic. A Quasistatic process ensures that the system will go through a sequence of states that are infinitesimally close to equilibrium.
7. In analysis of mechanical stresses, which of the following is the correct form of the 3D equation of stress equilibrium?
a) \(\frac{\partial \sigma x}{\partial x}+\frac{\partial \tau xy}{\partial y}+\frac{\partial \tau xz}{\partial z}\)=0
b) \( \frac{\partial \tau xy}{\partial x}+\frac{\partial \sigma y}{\partial y}+\frac{\partial \tau yz}{\partial z}\)=0
c)\( \frac{\partial \tau xz}{\partial x}+\frac{\partial \tau yz}{\partial y}+\frac{\partial \sigma z}{\partial x}\)=0
d) No such equation exists
View Answer
Explanation: For Cartesian problems in three dimensions, the stresses σx, τxy, and τxz act on the face normal to X-axis and the body is said to be in equilibrium if\(\frac{\partial \sigma x}{\partial x}+\frac{\partial \tau xy}{\partial y}+\frac{\partial \tau xz}{\partial z}\)=0 on that face.
8. In thermodynamics, what does the term open system refer to?
a) Control mass
b) Control volume
c) Control energy
d) Control temperature
View Answer
Explanation: In thermodynamics, an open system freely exchanges matter and energy with its surroundings. For instance, when you are boiling water in an open bowl on a stove, energy and matter are being transferred to the surroundings through steam. In this case, the volume remains constant making it a control volume process.
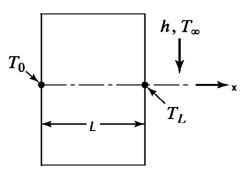
9. Consider the wall of a tank containing a hot liquid at a temperature T0, with an airstream passed on the outside, maintaining a wall temperature of TL at the boundary. From the figure shown,what is the boundary condition at X=0?
a) T=T0
b) T=T∞
c) T=TL
d) Data insufficient
View Answer
Explanation: For heat transfer problems, the boundary conditions are mainly of three types. They are specified temperature, specified heat flux (zero for insulated) and convection. From the figure shown, at X=0, the boundary condition specified is T=T0.
10. In thermodynamics, what is the name of a process with identical end states?
a) Cycle
b) Path
c) Phase
d) Either Path or phase
View Answer
Explanation: A thermodynamic cycle is a process with identical end states. A thermodynamic path is series of states or the path through which a system passes from an initial state of equilibrium to a final state of equilibrium. Phase is a quantity of matter that can be separated mechanically from a heterogeneous mixture.
11. For the given heat radiation equation, which of the following terminology is not correct?
q{x}n{x}+q{y}n{y}+q{z}n{z}=σε\(T_{\{s\}}^{\{4\}}\)-αq{r}
a) α is Stefan-Boltzmann constant
b) σ is the surface emission coefficient
c) ε is surface absorption coefficient
d) qr is heat generated per unit surface area
View Answer
Explanation: In the given equation,the first term in the RHS denotes radiated power (outgoing flow) and it has no sign before it. The second term has negative sign before it. Thus, qr denotes negative (incoming) heat flow per unit surface area. qr does not denote heat generated per unit surface area.
12. A system is said to be in thermodynamic equilibrium if it maintains which of the following equilibriums?
a) Mechanical and phase
b) Thermal and chemical
c) Thermal, mechanical and chemical
d) Thermal, phase, mechanical and chemical
View Answer
Explanation: Thermodynamic equilibrium describes a system whose properties will not change without some sort of outside interference. In other words, a system in thermodynamic equilibrium will not change unless something is added or subtracted from it. Thus, it should be under Thermal, mechanical and chemical equilibrium.
13. Which method is used to write the basic heat transfer equation in the following form using weighted residuals?
\(\int_v \left(\frac{\partial q_x}{\partial x}+\frac{\partial q_y}{\partial y}+\frac{\partial q_z}{\partial z}-Q+ \rho c \frac{\partial T}{\partial t}\right)\) Ni dV = 0
a) Galerkin method
b) Jacobi method
c) Rayleigh Ritz method
d) Delaunay method
View Answer
Explanation: Finite element equations are obtained using Galerkin method. Rayleigh Ritz method does not use interpolation functions, Ni whereas Galerkin method uses interpolation functions, Ni and thus, is used in FEM. Jacobi is used for Eigen value problems. Delaunay method is used to generate mesh for triangular elements.
Sanfoundry Global Education & Learning Series – Finite Element Method.
To practice Finite Element Method Question Bank, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Check Civil Engineering Books
- Apply for Mechanical Engineering Internship
- Practice Mechanical Engineering MCQs
- Practice Civil Engineering MCQs
- Check Finite Element Method Books
