This set of Enzyme Technology Quiz focuses on “Kinetics of Enzyme Catalysed Reaction – 2”.
1. What does the equation \(V_0=\frac{V_{max} [S]}{K_m+[S]}\) represent?
a) Lineweaver Burk equation
b) Miachelis Menten equation
c) Eadie-Hofstee plot equation
d) Hanes plot equation
View Answer
Explanation: The equation \(V_0=\frac{V_{max} [S]}{K_m+[S]}\) represents Miachelis Menten equation, where
V0 = initial velocity, Vmax = maximum rate, Km = Miachelis Menten constant and [S] = substrate concentration.
Line weaker Burk equation, Eadie-Hofstee plot equation and Hanes plot equation are derivatives of Miachelis Menten equation.
2. Which of the following is termed as catalytic efficiency?
a) Kcat
b) Km
c) Kcat/Km
d) Vmax
View Answer
Explanation: Kcat/Km is termed as catalytic efficiency. A high value indicates that the limiting factor of the overall reaction is the collision frequencies between enzyme and substrate molecule, whereas the low value indicates the equilibrium assumption status. Km and Kcat represent the Miachelis Menten constant and turn over number respectively. Vmax is the maximum rate of the reaction catalyzed by enzymes.
3. Which of the following equation is Eadie-Hofstee plot equation?
a) \(\frac{1}{V_0} = \frac{K_m}{V_{max}} \frac{1}{[S]}+\frac{1}{V_{max}}\)
b) \(V_0=\frac{V_{max} [S]}{K_m+[S]}\)
c) \(V_0=K_m \frac{V_0}{S_0}+V_{max}\)
d) \(\frac{[S_0]}{V_0} = \frac{1}{V_{max}} [S_0]+\frac{K_m}{V_{max}} \)
View Answer
Explanation: The Lineweaver Burk equation has its own limitations due to the following reasons:
The extrapolation of graph to determine the -1/Km reaches the edge of the paper such that, the accurate value is unable to calculate.
The -1/Km value calculated for low substrate concentrations are seemed to be inaccurate for most times.
The linearity of the graph for calculating the -1/Km value is not clear.
To overcome these, Eadie-Hofstee plot equation was designed. The equation is
\(V_0=K_m \frac{V_0}{S_0} + V_{max}\) where,
V0 = initial velocity, Vmax = maximum rate, Km = Miachelis Menten constant and [S] = substrate concentration.
4. The equation which is based on reciprocal form of Miachelis Menten equation at constant [Et] level is referred to as ______________
a) Eadie-Hofstee plot equation
b) Eisenthal and Cornish and Bowden plot equation
c) Lineweaver Burk equation
d) Hanes plot equation
View Answer
Explanation: To overcome certain limitation posed by various plots in determining Km and Vmax, the equation based on reciprocal form of Miachelis Menten equation is referred to as Eisenthal and Cornish and Bowden plot equation. The equation is given by \(\frac{V_{max}}{V_0} = \frac{K_m}{[S_0]}+1\) where,
V0 = initial velocity, Vmax = maximum rate, Km = Miachelis Menten constant and [S] = substrate concentration.
At constant V0 and S0, a plot of Vmax against Km will be linear.
5. ______________ is defined as an enzyme molecule’s efficiency to convert maximum substrate molecules into product per unit time.
a) Catalytic efficiency
b) Miachelis Menten constant
c) Turn over number
d) Vmax
View Answer
Explanation: Turn over number is defined as an enzyme molecule’s efficiency to convert maximum substrate molecules into product per unit time. Turn over number is given Kcat and it denotes the catalytic turn over events that happen per unit time. The turn over number for most enzymes ranges from 1 to 105 per second. Miachelis Menten constant is Km which is calculated as substrate concentration, which results in the half- maximal velocity of the enzymatic reaction. Kcat/Km is termed as catalytic efficiency. Vmax is referred to as maximum rate of an enzyme catalyzed reaction.
6. In the following plot, what does X represent?
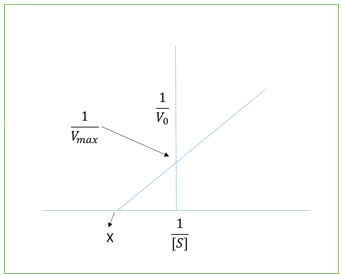
a) Vmax
b) Km/Vmax
c) –1/Km
d) Smax
View Answer
Explanation: The plot represents Lineweaver Burk plot which is devised from the equation, \(\frac{1}{V_0} = \frac{K_m}{V_{max}} \frac{1}{[S]}+\frac{1}{V_{max}}\) where,
V0 = initial velocity, Vmax = maximum rate, Km = Miachelis Menten constant and [S] = substrate concentration.
According to this plot, the slope gives Km/Vmax, the y intercept gives 1/Vmax and the X on the x-intercept gives -1/Km.
7. What does the following plot represents?

a) Miachelis Menten plot
b) Lineweaver Burk plot
c) Eadie-Hofstee plot
d) Hanes plot
View Answer
Explanation: Eadie-Hofstee plot is a graphical representation of enzyme kinetics in which reaction velocity is plotted as a function V/[S] which is based on the equation \(V_0=K_m \frac{V_0}{S_0} + V_{max}\) where,
V0 = initial velocity, Vmax = maximum rate, Km = Miachelis Menten constant and [S] = substrate concentration.
The above plot yields a straight line of slope –Km, a x-intercept Vmax/Km and an y-intercept Vmax.
8. A low Km value points out a low affinity of an enzyme towards its substrate.
a) True
b) False
View Answer
Explanation: Km is defined as the substrate concentration which, results in the half-maximal velocity of the enzyme reaction. The value of Km varies from one enzyme to other and also affected by certain conditions like pH, temperature, ionic strength, nature of solvent etc. Also, the values of Km can be different for the mutant forms of enzyme, isoenzyme and even for different substrates catalyzed by the same enzyme. A low value Km specifies, a high affinity of an enzyme towards its substrate i.e., at even low substrate concentration of substrate an enzyme can initiate the reaction and a high Km points out a low affinity of an enzyme towards its substrate. Hence the above statement is false.
9. Given an enzyme with Km of 0.25mM, at what substrate concentration will the velocity of the enzyme reach 1/2 of the Vmax? (Vmax = 100 mmol/s)
a) 0.5 mM
b) 0.25 mM
c) 0.75 mM
d) 25 mM
View Answer
Explanation: \(V_0 = \frac{1}{2} V_{max}\)
\(V_0 = \frac{1}{2}*100\)
V0 = 50 mmol/s
\(V_0 = \frac{V_{max} [S]}{K_m+[S]}\)
50 = \(\frac{100 [S]}{0.25+[S]}\)
\(\frac{1}{2} = \frac{[S]}{0.25+[S]}\)
0.25+[S]=2[S]
[S]=0.25 mM.
10. An enzyme with a Km of 5mM has a reaction rate of 100 mmol/min at substrate concentration of 0.25 mmol. What is the maximum reaction rate that this enzyme can achieve when its saturated with substrate?
a) 2100
b) 1500
c) 1900
d) 9000
View Answer
Explanation: Miachelis Menten equation is given by \(V_0=\frac{V_{max} [S]}{K_m+[S]}\)
\(V_{max} = \frac{V_0 (K_m+[S])}{[S]}\)
\(V_{max} = \frac{100 (5+0.25)}{0.25}\)
\(V_{max} = \frac{525}{0.25}\)
\(V_{max}\) = 2100 mmol/min.
Sanfoundry Global Education & Learning Series – Enzyme Technology.
To practice all areas of Enzyme Technology for Quizzes, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Apply for Biotechnology Internship
- Practice Biotechnology MCQs
- Check Biotechnology Books
- Check Enzyme Technology Books
- Apply for Chemical Engineering Internship
