This set of Enzyme Technology Multiple Choice Questions & Answers (MCQs) focuses on “Potentiometric Biosensors”.
1. Potentiometric biosensors uses _______________ in order to transduce the biological reaction into an electrical signal.
a) ion-selective electrodes
b) clark oxygen electrode
c) thermistors
d) colorimetric test strips
View Answer
Explanation: In potentiometric biosensors, ion-selective electrodes transduce biological reaction into an electrical signal. Clark oxygen electrode are used in amperometric biosensors, whereas colorimetric test strips are used in optical biosensors. Thermistors are used to measure the temperature difference in calorimetric biosensors.
2. Which of this is not true for ion-selective electrode?
a) Determination of electrical potential at very high impedance
b) Allowing zero current flow effectively
c) Increasing the heat output
d) Causing no interference with the reaction
View Answer
Explanation: Increasing the heat output is true in case of thermistors that are used in calorimetric biosensors. For ion-selective electrodes, the following statements are true:
• Determination of electrical potential at very high impedance,
• Allowing zero current flow effectively,
• Causing no interference with the reaction.
3. What is represented in the following diagram?
a) Amperometric biosensor
b) Piezo-electric biosensor
c) Potentiometric biosensor
d) Calorimetric biosensor
View Answer
Explanation: The diagram below represents a simple potentiometric biosensor with its parts.
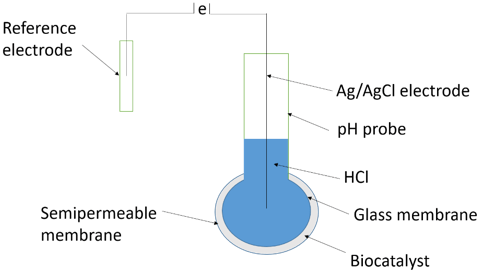
A simple potentiometric biosensor has a semi-permeable membrane surrounding the biocatalyst which is entrapped next to the active glass membrane of a pH probe. The electrical potential generated between the internal Ag/AgCl electrode bathed in dilute HCl and an external reference electrode is given by ‘e’.
4. Which of these is not an ion-selective electrode?
a) Thermistors
b) Glass electrodes
c) Glass pH electrodes
d) Solid-state electrodes
View Answer
Explanation: Thermistors are used in calorimetric biosensor to measure the heat generated during the reaction. Hence it is not an ion-selective electrode. Glass electrodes, glass pH electrodes and solid-state electrodes are types ion-selective electrodes that are used in the potentiometric biosensor.
5. ______________ is one in which the sensing element is a very thin hydrated glass membrane.
a) Solid-state electrode
b) Glass electrode
c) Thermistor
d) Glass pH electrode
View Answer
Explanation: Glass electrode is one in which the sensing element is a very thin hydrated glass membrane which generates a transverse electrical potential. The electrical generated is due to concentration-dependent competition between the cations for specific binding sites. In glass pH electrode, the diffusion of gas through the membrane causes a change in pH between the membrane and the electrode, which can be determined later. Solid-state electrodes has thin membrane of a specific ion conductor made from a mixture of silver sulphide and a silver halide. Thermistor is used to measure the heat generated.
6. Which of the following ion-selective electrode consist of gas permeable membrane?
a) Glass electrode
b) Solid-state electrode
c) Glass pH electrode
d) Clark oxygen electrode
View Answer
Explanation: Glass pH electrode consists of gas permeable membrane selective for CO2, NH3 or H2S. The diffusion of the gas through this membrane causes a change in pH of a sensing solution between the membrane and the electrode which is then determined. Glass electrode has a hydrated glass membrane which generates transverse electric potential. Solid-state electrodes has thin membrane of a specific ion conductor made from a mixture of silver sulphide and a silver halide. Clark oxygen electrode is used in amperometric biosensor.
7. ____________ is one in which has a thin membrane of a specific ion conductor made from a mixture of silver sulphide and a silver halide.
a) Glass electrode
b) Solid-state electrode
c) Glass pH electrode
d) Clark oxygen electrode
View Answer
Explanation: In solid state electrode, the glass membrane is replaced with a thin membrane of a specific ion conductor made from a mixture of silver sulphide and a silver halide. Glass electrode and Glass pH electrode has hydrated glass membrane and gas permeable membrane respectively. Clark oxygen electrode is used in amperometric biosensors.
8. What does ‘X’ represent in the following reaction?
Penicillin \(\underset{X}{\rightarrow}\) Penicilloic acid + H+
a) Asparginase
b) Lipase
c) Urease
d) Penicillinase
View Answer
Explanation: In the above reaction, ‘X’ is penicillinase which is an enzyme that acts on penicillin producing hydrogen ions which can be measured by using potentiometric biosensor. Asparaginase acts on asparagine releases NH4+ ions which can be measured. Urease acts on urea releasing H+ ions, whereas lipases acts on lipids and release hydrogen ions.
9. What does ‘Y’ represent in the following reaction?
Amygdalin + 2H2O \(\underset{X}{\rightarrow}\) 2 Glucose + benzaldehyde + H+ + CN–
a) Peroxidase
b) L-aminoacid oxidase
c) β – glucosidase
d) Urease
View Answer
Explanation: In the above reaction, ‘Y’ represent enzyme β – glucosidase which acts amygdalin releasing glucose, benzaldehyde, cyanide ions and hydrogen ions which can be measured using potentiometric biosensor. Peroxidases acts on hydrogen peroxide releasing iodine ions and water. L-aminoacid oxidases oxidizes L-aminoacids to ketoacids, ammonia ions and hydrogen peroxide. Urease acts on urea releasing different products.
10. What does the following diagram represent?
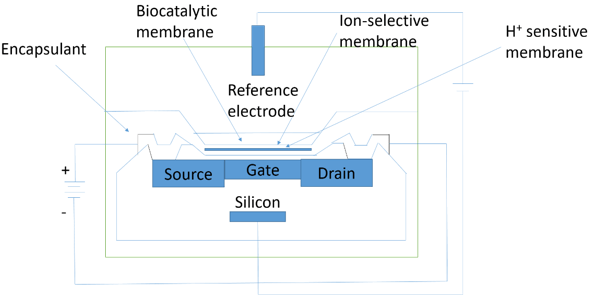
a) Potentiometric biosensor
b) ENFET
c) Glass pH electrode
d) Piezo-electric biosensor
View Answer
Explanation: In the above diagram, ENFET is represented. ENFET means enzyme-linked field effect transistors. The main body of the biosensor is a p-type silicon chip with two n-type silicon areas. The chip is insulated by a thin layer of silica which forms the gate of the FET. Above the gate, there is an equally thin layer of H+-sensitive material, a protective ion selective membrane, the biocatalyst and the analyte solution, which is separated by an inert encapsulating polyimide photopolymer. When a potential is applied between the electrodes, a current flows through the FET dependent upon the positive potential detected at the ion-selective gate and its consequent attraction of electrons into the depletion layer.
Sanfoundry Global Education & Learning Series – Enzyme Technology.
To practice all areas of Enzyme Technology, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Check Biotechnology Books
- Apply for Biotechnology Internship
- Practice Biotechnology MCQs
- Practice Chemical Engineering MCQs
- Check Enzyme Technology Books
