This set of Enzyme Technology Multiple Choice Questions & Answers (MCQs) focuses on “Amperometric Biosensors”.
1. Which of the following biosensors function by the production of a current when a potential is applied between two electrodes?
a) Calorimetric biosensor
b) Potentiometric biosensor
c) Optical biosensor
d) Amperometric biosensor
View Answer
Explanation: Amperometric biosensor function by the production of a current when a potential is applied between two electrodes. They generally have response times, dynamic ranges and sensitivities similar to the potentiometric biosensors, which makes use of ion-selective electrodes in order to transduce the biological reaction into an electrical signal. Optical biosensors involve determining changes in light absorption between the reactants and products of a reaction or measuring the light output. Calorimetric biosensor involves measuring the heat generated during a reaction using thermistors.
2. The simplest amperometric biosensor is ____________
a) glass electrode
b) thermistor
c) clark oxygen electrode
d) solid-state electrode
View Answer
Explanation: The simplest amperometric biosensor is clark oxygen electrode, which consists of a platinum cathode at which oxygen is reduced and a silver/silver chloride reference electrode. Glass electrodes is an ion-selective electrode which has very thin hydrated glass membrane which generates a transverse electrical potential. Thermistors are used to measure the temperature changes in a calorimetric biosensor. Solid-state electrode is an ion-selective electrode which has thin membrane of a specific ion conductor made from a mixture of silver sulphide and a silver halide.
3. The following reaction occurs at __________
4Ag0 + 4Cl– → 4AgCl + 4e–
a) Pt cathode
b) Ag cathode
c) Ag anode
d) Pt anode
View Answer
Explanation: Amperometric biosensor consists of a platinum cathode at which oxygen is reduced and a silver/silver chloride reference electrode. The above reaction occurs at Ag anode releasing 4e– which are then utilized by oxygen at Pt cathode reducing the oxygen concentration to zero. Pt anode and Ag cathode does not exist and hence not used on amperometric biosensor.
4. Where does the following reaction occur?
O2 + 4H+ + 4e– → 2H2O
a) Pt cathode
b) Pt anode
c) Ag anode
d) Ag cathode
View Answer
Explanation: Pt anode and Ag cathode does not exist. At Pt cathode, reduction occurs of oxygen to water by involving the 4 electrons released at Ag anode. Oxidation occurs at Ag anode and reduction at Pt cathode. These reactions are involved in amperometric type of biosensor.
5. Which of the following biosensor determines glucose concentrations by the use of an immobilized glucose oxidase membrane?
a) Optical biosensor
b) Amperometric biosensor
c) Calorimetric biosensor
d) Potentiometric biosensor
View Answer
Explanation: A typical example of amperometric biosensor is determination of glucose concentrations by the use of an immobilised glucose oxidase membrane. An example of optical biosensor is luciferase which is used to detect the presence of bacteria in food or clinical samples. Glucose analysis using glucose oxidase by co-immobilization with catalase is a typical example of measurement using calorimetric biosensor. Potentiometric biosensors are used to determine asparginase activity of measuring the release of NH4+ ions.
6. Which of the biosensor is presented in the diagram?
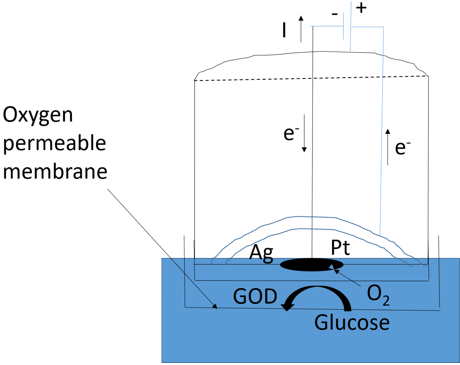
a) Immunosensor
b) Piezo-electric biosensor
c) Potentiometric biosensor
d) Optical biosensor
View Answer
Explanation: In the above diagram, simplest form amperometric biosensor is shown. In amperometric biosensor, an electric potential is applied between the central platinum cathode and the annular silver anode in the presence of saturated solution of KCl which generates a current. This electrode compartment is separated from the Glucose oxidase (biocatalyst) by a thin plastic membrane that is permeable only to oxygen. The analyte solution is separated from the biocatalyst by another membrane which is permeable to the substrate and product.
7. Use of mediators in amperometric biosensor is required.
a) True
b) False
View Answer
Explanation: In amperometric biosensor, there is an efficient reduction of oxygen at the surface of the cathode causes the oxygen concentration there to be effectively zero. But the major problem in this case is their dependence on the dissolved oxygen concentration. To overcome this, mediators may be used which transfers the electrons directly to the electrode bypassing the reduction of the oxygen co-substrate. Hence the above statement is true.
8. Which of these properties is not true for mediators?
a) Mediators must react rapidly with a reduced form of enzyme
b) Mediators must be sufficiently soluble in oxidized and reduced forms
c) The reduced form of the mediator should not readily react with oxygen
d) Increasing the heat output
View Answer
Explanation: The properties which are true for mediators used in amperometric biosensor are as follows:
• Mediators must react rapidly with reduced form of enzyme.
• Mediators must be sufficiently soluble in oxidized and reduced forms.
• The reduced form of the mediator should not readily react with oxygen.
• The over potential for the regeneration of the oxidized mediator should be low and independent of pH.
Increasing the heat output is true for thermistors used in calorimetric biosensor.
9. Which of the following represents a commonly used family of mediators?
a) Pyruvate kinase
b) Hexokinase
c) Ferrocenes
d) Lactate oxidase
View Answer
Explanation: Ferrocenes represent a commonly used family of mediators. An extreme case of this amplification is the detection of ADP. ADP is the added analyte and excess glucose, phosphoenol pyruvate, NADH and oxygen are present to ensure maximum reaction. Four enzymes such as hexokinase, pyruvate kinase, lactate dehydrogenase and lactate oxidase are co-immobilised within the packed bed reactor.
10. Amperometric biosensors for flavo-oxidase enzymes illustrates three generations in the development of a biosensor.
a) True
b) False
View Answer
Explanation: Amperometric biosensors for flavo-oxidase enzymes illustrates three generations in the development of a biosensor which are as follows:
• 1st generation electrode: utilizes H2O2 produced by the reaction.
• 2nd generation electrode: utilizes a mediator to transfer the electrons, produced by the reaction, to the electrode.
• 3rd generation electrode: utilizes the electrons produced by the reaction.
Hence the above statement is true.
11. What is represented by the following equation?
I = nFAvA
a) Frequency
b) Current
c) Electrical resistance
d) Electrical potential
View Answer
Explanation: The equation represents current produced by amperometric biosensors is related to the rate of reaction by the expression:
I = nFAvA where,
n = the number of electrons transferred, A = electrode area, vA = rate of reaction, and F = Faraday.
Sanfoundry Global Education & Learning Series – Enzyme Technology.
To practice all areas of Enzyme Technology, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Practice Chemical Engineering MCQs
- Check Chemical Engineering Books
- Check Biotechnology Books
- Check Enzyme Technology Books
- Practice Biotechnology MCQs
