This set of Engineering Physics written test Questions & Answers focuses on “Interaction of External Energy with the Atomic Energy States”.
1. Photons emitted by spontaneous emission are __________
a) Coherent and Monochromatic
b) Non-coherent and monochromatic
c) Coherent and Non-Monochromatic
d) Non-Coherent and Non-monochromatic
View Answer
Explanation: Spontaneous emission is the one when an atom undergoes transition to a lower energy state emitting a photon, without external stimulation. In this process, the emitted photon is non-coherent and non-monochromatic.
2. In Stimulated Absorption, what is the lifetime of atoms ground state?
a) 1 second
b) 1 minute
c) 1 hour
d) Infinity
View Answer
Explanation: At the ground state, the atoms are perfectly stable. They are under no excessive force that might lead to become unstable. All the forces are balanced. Thus, as the atom is stable in ground state, it’s lifetime is infinity.
3. Phonons are __________
a) Quanta of energy
b) Quanta of light waves
c) Quanta of sound waves
d) Quanta of heat
View Answer
Explanation: Phonons are the quanta of sound waves. When energy is provided, the lattice absorbs energy and gets excited to a higher state. When it de-excites to ground state, it releases radiation in sound-wave region, known as phonons.
4. The frequency of incident photon so that the atom makes a transition from E1 to E2 should be __________
a) E2 – E1
b) E2 – E1/c
c) E2 – E1/h
d) E2 – E1/λ
View Answer
Explanation: For an atom to make a transition from E2 to E1, the energy of the incident photon should be E2 – E1. Therefore, hv = E2 – E1. Hence, v = E2 – E1/h.
5. What is the general lifetime of an atom in an exited state?
a) 10-10
b) 10-8
c) 10-6
d) 10-4
View Answer
Explanation: The excited state is an unstable state. The exited atom tends to jump back to the ground state, where it will be stable. Therefore, it’s lifetime in the exited state is 10-8s.
6. The output of a laser has pulse duration of 20 ms and average output power of 1.75 W per pulse. How much energy is released per pulse if wavelength is 5890 Å?
a) 0.011 J
b) 0.024 J
c) 0.035 J
d) 0.047 J
View Answer
Explanation: As we know, Energy = Power X Time
= 1.75 W X 20 X 10-3 s
= 0.035 J.
7. Laser light from a 7mW source of aperture diameter 1.5 cm and wavelength 5000 Å is focused by a lens of focal length 10 cm. The intensity of the image is __________
a) 3 X 107 Wm-2
b) 4 X 107 Wm-2
c) 5 X 107 Wm-2
d) 6 X 107 Wm-2
View Answer
Explanation: Area of the image = \(\frac{\pi\lambda^2f^2}{a^2}\)
λ = 5000 Å = 5.0 X 10-7 m, f = 0.1 m
a = 1.5/2 cm = 0.75 X 10-2 m
Putting in the formula we get, Area = 1.4 X 10-10 m2
Intensity = Power/Area
= 7 X 10-3 W/ 1.4 X 10-10 m2
= 5 X 107 Wm-2.
8. For an ordinary light source, the coherence time t = 10-10s. The degree of Monochromaticity for a wavelength of 6000 Å is ___________
a) 0.1 X 10-4
b) 0.2 X 10-4
c) 0.3 X 10-4
d) 0.4 X 10-4
View Answer
Explanation: Coherence time, t = 10-10 s
Therefore, Δv = 1/t = 1010 Hz
Now, λo = 5000 Å, vo = 5.0 X 1014 Hz
Monochromaticity = \(\frac{Δv}{v_0}\)
= 0.2 X 10-4.
9. In Stimulated emission, the emitted photons are _____________
a) Coherent and Monochromatic
b) Non-coherent and monochromatic
c) Coherent and Non-Monochromatic
d) Non-Coherent and Non-monochromatic
View Answer
Explanation: Contrary to spontaneous emission, the emitted photon in stimulated emission are coherent and monochromatic. That is why this process is used for the production of LASERS.
10. The following graph is pictorial representation of _____________
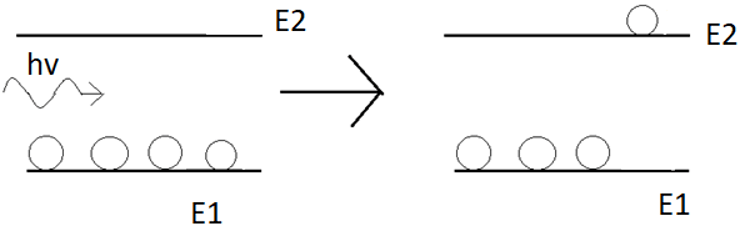
a) Spontaneous emission
b) Spontaneous Absorption
c) Stimulated emission
d) Stimulated Absorption
View Answer
Explanation: The diagram shows that when a photon from the incident radiation, having energy E2 – E1, interacts with the atom in the ground state, the atom gets exited to a higher energy state. This is known as stimulated absorption.
Sanfoundry Global Education & Learning Series – Engineering Physics.
To practice all written questions on Engineering Physics, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
