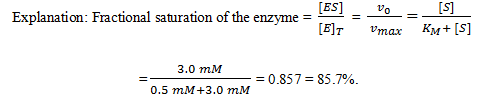This set of Bioprocess Engineering test focuses on “General Reaction Kinetics for Biological Systems”.
1. Which order represents Michelis-Menten kinetics?
a) First-Second order
b) Zero-First order
c) Zero-Second order
d) Second order
View Answer
Explanation: The kinetics of many biological reactions are either zero-order, first-order or a combination of these called Michaelis-Menten kinetics. The reaction order depends on the relative size of the two terms in the denominator. At low substrate concentration [S] <<KM, under these conditions the reaction rate varies linearly with substrate concentration [S] – First order kinetics. However at higher [S] with [S] >> KM, the reaction becomes independent of [S] – Zero order kinetics.
2. The relationship between reaction rate and reactant concentration is of which order from the following relation?
rA = k1 CA
a) Zero order
b) First order
c) Zero-First order
d) Second order
View Answer
Explanation: If a reaction obeys first-order kinetics, the relationship between reaction rate and reactant concentration is as follows:
rA = k1 CA
where rA is the volumetric rate of reaction and k1 is the first- order rate constant with dimensions T-1.
3. The following enzyme reaction is applicable for which order?

a) Zero order
b) Zero-First order
c) Zero-Second order
d) First order
View Answer
Explanation: The kinetics of most enzyme reactions are reasonably well represented by the Michaelis-Menten equation, where rA is the volumetric rate of reaction, CA is the concentration of reactant A, Vmax is the maximum rate of reaction at infinite reactant concentration, and Km is the Michaelis constant for reactant A. Vmax has the same dimensions as rA; Km has the same dimensions as CA.
4. Refer to Q3, and estimate the unit of Km?
a) kg mol m-2
b) kg mol
c) kg mol m-1
d) kg mol m-3
View Answer
Explanation: Km is the Michaelis constant for reactant A. Km has the same dimensions as CA. Typical units for Vmax are kgmol m-3 s-I; typical units for Km are kgmolm-3.
5. What do you mean by the “Turn- over number”?
a) Occurrence of saturation
b) Conversion of amount of substrate into product
c) Conversion of amount of product into substrate
d) Enzyme unbound with substrate
View Answer
Explanation: When all enzyme is bound to substrate kcat, the turnover number, is the maximum number of substrate molecules converted to product per enzyme molecule per second. Further addition of substrate does not increase the rate which is said to be saturated.
6. High substrate concentration follows which order?
a) Zero order
b) First order
c) Zero- Second order
d) Second order
View Answer
Explanation: v ≈ vmax
Therefore, at high substrate concentrations, the reaction rate approaches a constant value independent of substrate concentration; in this concentration range, the reaction is essentially zero order with respect to the substrate.
7. Convert 35°C into K.
a) 300.08 K
b) 305.18 K
c) 308.15 K
d) 315.18 K
View Answer
Explanation: Convert temperatures to degrees Kelvin (K) using this equation:
T(K) = T(°C) + 273.15
T = 35°C = 308.15 K.
8. Convert 35 K into °C.
a) 230.18
b) 238.15
c) -230.18
d) -238.15
View Answer
Explanation: Convert degree Kelvin (K) into temperatures using this equation:
T(°C) = T(K) – 273.15
= -238.15°C.
9. Catalytic efficiency is defined as__________
a) kcat/KM
b) KM/kcat
c) Km/k0
d) Km/k1
View Answer
Explanation: The constant kcat/KM (catalytic efficiency) is a measure of how efficiently an enzyme converts a substrate into product.
10. Unbinding of Enzyme- Substrate complex increases the reaction rate.
a) True
b) False
View Answer
Explanation: The rate of an enzymatic reaction will increase as substrate concentration increases, and that increased unbinding of enzyme-substrate complexes will decrease the reaction rate.
11. Linear inhibition is sometimes called as ____________
a) Complete inhibition
b) Partial inhibition
c) Incomplete inhibition
d) Mixed inhibition
View Answer
Explanation: Linear inhibition is sometimes called complete inhibition, and the contrasting term partial inhibition is sometimes used for a type of non-linear inhibition in which saturation with inhibitor does not decrease the rate to zero. These latter terms are discouraged because they can be misleading, implying, for example, that the rate may indeed be decreased to zero in ‘complete inhibition’ at non-saturating concentrations of inhibitor.
12. The rate limiting step of Michaelis Menten kinetics is _________
a) Complex formation step
b) Non-complex formation step
c) Complex dissociation step
d) Non- complex dissociation step
View Answer
Explanation: The rate limiting step in the enzyme catalyzed transformation of substrate S into product P is the breakdown of the ES complex. The rate-limiting step is usually the product formation step.
13. For a given enzyme catalyzed reaction, the Michaelis constant is 0.5 mM and the substrate concentration is 3.0 mM. What is the fractional saturation of the enzyme under these conditions?
a) 80.57%
b) 85.5%
c) 85.7%
d) 80.75%
View Answer
14. In a particular enzyme-catalyzed reaction, Vmax = 0.2 mol/sec and Km = 5 mM. Assume the enzyme shows standard Michaelis-Menten kinetics. What is the rate of the reaction when [S] = 10 mM?
a) 0.133
b) 0.331
c) 0.233
d) 0.332
View Answer
Explanation: v = Vmax[S] / (Km + [S])
= 0.2×10 / (5+10)
= 0.133.
15. In a particular enzyme-catalyzed reaction, Vmax = 0.2 mol/sec and Km = 6 mM. Assume the enzyme shows standard Michaelis-Menten kinetics. What is the rate of the reaction when [S] = 20 mM?
a) 0.155
b) 0.156
c) 0.153
d) 0.152
View Answer
Explanation: v = Vmax[S] / (Km + [S])
= 0.2×20 / (6+20)
= 0.153.
Sanfoundry Global Education & Learning Series – Bioprocess Engineering.
To practice all areas of Bioprocess Engineering for tests, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Check Bioprocess Engineering Books
- Apply for Biotechnology Internship
- Check Biotechnology Books
- Practice Biotechnology MCQs