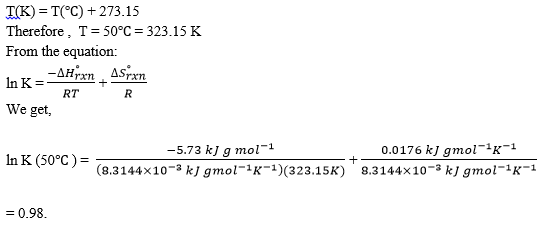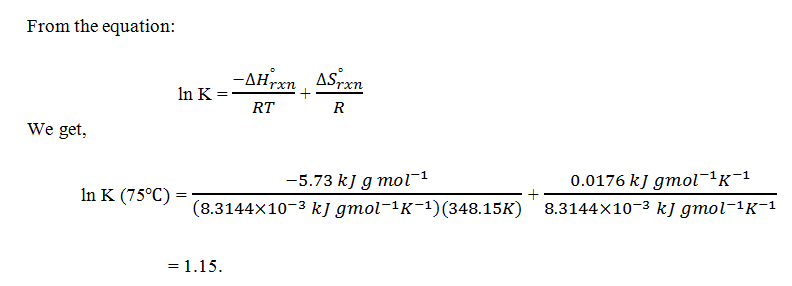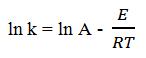This set of Bioprocess Engineering Multiple Choice Questions & Answers (MCQs) focuses on “Basic Reaction Theory”.
1. Which type of catalysis reaction defines cell growth?
a) Tandem catalysis
b) Surface catalysis
c) Homo catalysis
d) Autocatalysis
View Answer
Explanation: Cell growth is an autocatalytic reaction: this means that the catalyst is a product of the reaction. The performance of catalytic reactions is characterized by variables such as the reaction rate and yield of product from substrate. In bioreactors, the growth is autocatalytic in that the more cells you have, the greater the growth rate.
2. According to reaction theory, Reaction kinetics is concerned with how far the reaction can proceed.
a) True
b) False
View Answer
Explanation: Reaction theory has two fundamental parts: reaction thermodynamics and reaction kinetics. Reaction thermodynamics is concerned with how far the reaction can proceed; no matter how fast a reaction is, it cannot continue beyond the point of chemical equilibrium. On the other hand, reaction kinetics is concerned with the rate at which reactions proceed.
3. The equilibrium constant (K) depends on __________
a) Pressure
b) pH
c) Temperature
d) Density
View Answer
Explanation: The value of K depends on temperature as follows:
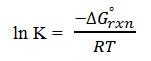
where, ΔG°rxn is the change in standard free energy per mole of A reacted, R is the ideal gas constant and T is absolute temperature.
4. For exothermic reactions, which condition is applicable for equilibrium constant K?
a) K decreases with increasing temperature
b) K decreases with decreasing temperature
c) Positive ΔH°rxn with increasing temperature; K increases
d) Negative ΔH°rxn with increasing temperature; K increases
View Answer
Explanation: For exothermic reactions with negative ΔH°rxn, K decreases with increasing temperature. For endothermic reactions and positive ΔH°rxn , K increases with temperature.
5. Glucose isomerase is used extensively in the USA for production of high-fructose syrup. The reaction is ___________
Glucose ⇄ Fructose
ΔH°rxn for this reaction is 5.73 kJ gmol-l; ΔS°rxn is 0.0176 kJ gmol-l K-l.
Calculate the equilibrium constants at 50°C. (Given R = 8.3144J gmol-l K-l =8.3144×10-3 kJ gmol-lK-l).
a) 0.95
b) 0.94
c) 0.96
d) 0.98
View Answer
6. Refer to Q5 and, calculate the same for equilibrium constant at 75°C.
a) 1.15
b) 1.16
c) 1.18
d) 1.17
View Answer
7. Refer to Q5 and Q6, and estimate that whether 75°C is desirable to operate?
a) True
b) False
View Answer
Explanation: K in 50°C is 0.98 and K in 75°C is 1.15.As K increases, the fraction of fructose in the equilibrium mixture increases. Therefore, from an equilibrium point of view, it is more desirable to operate the reactor at 75°C. However, other factors such as enzyme deactivation at high temperatures should also be considered.
8. Which of the following is not a reversible reaction?
a) Formation of NH3 rate
b) Decomposition of NH3 rate
c) Decomposition of potassium chlorate
d) Reaction of gaseous hydrogen with iodine vapors
View Answer
Explanation: Chemical reactions in which the products thus formed during a chemical change do not combine and produce back the reactants are better known as irreversible reactions. Decomposition of potassium chlorate into potassium chloride and oxygen after heating.
In the above reaction however, the products are not combining to form back the potassium chlorate. In case of physical and chemical processes which are irreversible in nature, the changes are found to be uni direction and the processes is completed.
9. The irreversible reaction takes place in the closed vessel.
a) True
b) False
View Answer
Explanation: For irreversible reaction Gibbs free energy change will be less than zero ΔG < 0 is the condition for the irreversible reaction. Irreversible reaction normally takes place in an open vessel.
10. What is the unit of the reaction yield?
a) gmol gmol
b) gmol2
c) gmolK-2
d) gmol / gmol
View Answer
Explanation: The theoretical yield is the amount predicted by a stoichiometric calculation based on the number of moles of all reactants present. This calculation assumes that only one reaction occurs and that the limiting reactant reacts completely.
11. “The rate per quantity of enzyme or cells involved in the reaction”, this is applicable to which type of reaction rate?
a) Total rate
b) Specific rate
c) Variable rate
d) Volumetric rate
View Answer
Explanation: In specific rate, biological reactions involve enzyme and cell catalysts. Because the total rate of conversion depends on the amount of catalyst present, it is sometimes useful to specify reaction rate as the rate per quantity of enzyme or cells involved in the reaction.
12. How do we measure the concentration of the enzyme?
a) In units of mass
b) In units of time
c) In units of activity
d) In units of volume
View Answer
Explanation: The mass of a particular enzyme added to a reaction is rarely known; most commercial enzyme preparations contain several components in unknown and variable proportions depending on the batch obtained from the manufacturer. To overcome these difficulties, enzyme quantity is often expressed as units of activity measured under specified conditions. One unit of enzyme is usually taken as the amount which catalyses conversion of 1μmol substrate per minute at the optimal temperature, pH and substrate concentration.
13. Total, volumetric and specific productivities are different.
a) True
b) False
View Answer
Explanation: Total, volumetric and specific productivities are interrelated concepts in process design. For example, high total productivity could be achieved with a catalyst of low specific activity if the reactor is loaded with a high catalyst concentration. If this is not possible, the volumetric productivity will be relatively low and a larger reactor is required to achieve the desired total productivity.
14. The conditions of rate of reaction from the following is _______
a) Reactions proceeds with the increase of reactants
b) Reactions decrease with the decrease of reactants
c) Reactions increase with decrease in temperature
d) Reactions increase with increase in temperature
View Answer
Explanation: As reactions proceed, the concentrations of reactants decrease. In general, rate of reaction depends on reactant concentration so that the specific rate of conversion decreases simultaneously. Reaction rate also varies with temperature; most reactions speed up considerably as the temperature rises.
15. Variation of the rate constant k with temperature is described by which equation?
a) Arrhenius equation
b) Monod equation
c) Kirchoff’s equation
d) Newton’s second law of thermodynamics
View Answer
Explanation: Temperature has a significant kinetic effect on reactions. Variation of the rate constant k with temperature is described by the Arrhenius equation:
k = A e-E/RT
Where, k is the rate constant, A is the Arrhenius constant or frequency factor, E is the activation energy for the reaction, R is the ideal gas constant, and T is absolute temperature. According to the Arrhenius equation, as T increases, k increases exponentially.
Taking the natural logarithm of both sides of equation:
Thus, a plot of ln k versus 1/T gives a straight line with slope -E/R . For many reactions the value of E is positive and large, indicating a rapid increase in reaction rate with temperature.
Sanfoundry Global Education & Learning Series – Bioprocess Engineering.
To practice all areas of Bioprocess Engineering, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
- Check Biotechnology Books
- Apply for Biotechnology Internship
- Check Bioprocess Engineering Books
- Practice Biotechnology MCQs