This set of Basic Chemical Engineering Multiple Choice Questions & Answers (MCQs) focuses on “Material Balances Problems Involving Multiple Units”.
1. What is the value of P in the following process?

a) 5 mole
b) 10 mole
c) 15 mole
d) 20 mole
View answer
Explanation: 10 + 20 = P + 10 + 5, => P = 15 mole.
2. What is the value of P in the following process?
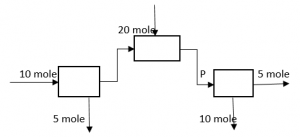
a) 5 mole
b) 15 mole
c) 25 mole
d) 35 mole
View answer
Explanation: 10 + 20 = 5 + P, => P = 25 mole.
3. What is the value of P in the following process?
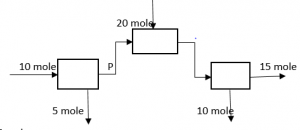
a) 5 mole
b) 10 mole
c) 15 mole
d) 20 mole
View answer
Explanation: 10 = P + 5, => P = 5 mole.
4. What is the value of F*P in the following process?
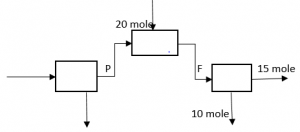
a) 25
b) 125
c) 150
d) 225
View answer
Explanation: F = 15 + 10 = 25 mole, P + 20 = F = 25, => P = 5 mole, => F*P = 125.
5. What is the value of H/P in the following process, if 2P + H = 16?
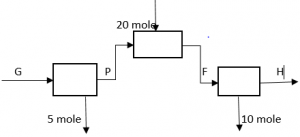
a) 1
b) 3
c) 6
d) 8
View answer
Explanation: P + 20 = H + 10, => H – P = 10, solving both equations, we get H = 12, P = 2, => H/P = 6.
Answer the following questions 6 – 10 for the diagram.
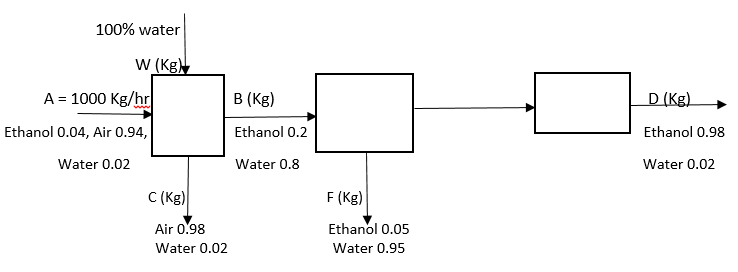
6. What is the value of B?
a) 100 Kg/hr
b) 200 Kg/hr
c) 300 Kg/hr
d) 400 Kg/hr
View answer
Explanation: Material balance for ethanol: 0.04*1000 = 0.2*B, => B = 200 Kg/hr.
7. What is the value of C?
a) 400 Kg/hr
b) 689.5 Kg/hr
c) 819.6 Kg/hr
d) 959.2 Kg/hr
View answer
Explanation: Material balance for air: 0.94*1000 = 0.98*C, => C = 959.2 Kg/hr.
8. What is the value of W?
a) 159.2 Kg/hr
b) 281.3 Kg/hr
c) 465.7 Kg/hr
d) 633.8 Kg/hr
View answer
Explanation: A + W = B + C, => 1000 + W = 200 + 959.2, => W = 159.2 Kg/hr.
9. What is the value of F?
a) 98.5 Kg/hr
b) 131.1 Kg/hr
c) 167.7 Kg/hr
d) 256.4 Kg/hr
View answer
Explanation: Material balance for ethanol: 200*0.2 = F*0.05 + D*0.98, => 5F + 98D = 4000, material balance for water: 200*0.8 = F*0.95 + D*0.02, => 95F + 2D = 16000, Solving both equations we get F = 167.7 Kg/hr.
10. What is the value of D?
a) 32.3 Kg/hr
b) 49.1 Kg/hr
c) 85.6 Kg/hr
d) 105.3 Kg/hr
View answer
Explanation: B = F + D, => 200 = 167.7 + D, => D = 32.3 Kg/hr.
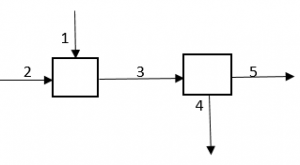
11. How many independent equations are possible, if only one component exists in each stream?
a) 1
b) 2
c) 3
d) 4
View answer
Explanation: For whole process: 1 + 2 = 4 + 5, for first reactor: 1 + 2 = 3, for second reactor: 3 = 4 + 5, but if we equate second and third equation we get the first one, so there are 2 independent equations.
12. How many independent equations are possible, if two components are involved in each of streams?

a) 1
b) 2
c) 3
d) 4
View answer
Explanation: It will be just the double of the number of independent equations if there was only one component.
13. 1 has pure A and 2 has pure B and 3, 4 and 5 has mixture of A and B, how many independent equations are possible?

a) 1
b) 2
c) 3
d) 4
View answer
Explanation: It will be just twice the number when there is only one component.
14. How many independent equations are possible if each stream contains mixture of A, B and C?

a) 1
b) 3
c) 6
d) 8
View answer
Explanation: It will be just thrice the number when there was only one component.
15. How many independent equation are possible if 1 has pure A, 2 has mixture of B and C, 3 has mixture of A, B and C, 4 has pure C, 5 has mixture of A and B.

a) 1
b) 3
c) 6
d) 9
View answer
Explanation: It will be just thrice the number when there was only one component.
Sanfoundry Global Education & Learning Series – Basic Chemical Engineering.
To practice all areas of Basic Chemical Engineering, here is complete set of 1000+ Multiple Choice Questions and Answers.
If you find a mistake in question / option / answer, kindly take a screenshot and email to [email protected]
